SECTION 1. IDENTIFICATION
Product Name: Barium Diphenylamine Sulfonate
Product Number: All applicable American Elements product codes, e.g. BADPH-SUNAT-01-LIQ
CAS #: 1300-92-1
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Physical hazards Not classified.
Health hazards Not classified.
Environmental hazards Not classified.
OSHA defined hazards Not classified.
Label elements
Hazard symbol None.
Signal word None.
Hazard statement The mixture does not meet the criteria for classification.
Precautionary statement
Prevention Observe good industrial hygiene practices.
Response Wash hands after handling.
Storage Store away from incompatible materials.
Disposal Dispose of waste and residues in accordance with local authority requirements.
Hazard(s) not otherwise
classified (HNOC)
None known.
Supplemental information None.
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Mixtures
*Designates that a specific chemical identity and/or percentage of composition has been withheld as a trade secret.
SECTION 4. FIRST AID MEASURES
Inhalation Move to fresh air. Call a physician if symptoms develop or persist.
Skin contact Wash off with soap and water. Get medical attention if irritation develops and persists.
Eye contact Rinse with water. Get medical attention if irritation develops and persists.
Ingestion Rinse mouth. Get medical attention if symptoms occur.
Most important
symptoms/effects, acute and
delayed
Direct contact with eyes may cause temporary irritation.
Indication of immediate
medical attention and special
treatment needed
General information Ensure that medical personnel are aware of the material(s) involved, and take precautions to
protect themselves.
SECTION 5. FIREFIGHTING MEASURES
Suitable extinguishing media Water fog. Foam. Dry chemical powder. Carbon dioxide (CO2).
Unsuitable extinguishing
media
Do not use water jet as an extinguisher, as this will spread the fire.
Specific hazards arising from
the chemical
During fire, gases hazardous to health may be formed.
Special protective equipment
and precautions for
firefighters
Self-contained breathing apparatus and full protective clothing must be worn in case of fire.
Fire fighting
equipment/instructions
Move containers from fire area if you can do so without risk.
Specific methods Use standard firefighting procedures and consider the hazards of other involved materials.
General fire hazards No unusual fire or explosion hazards noted.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions,
protective equipment and
emergency procedures
Keep unnecessary personnel away. For personal protection, see section 8 of the SDS.
Methods and materials for
containment and cleaning up
Large Spills: Stop the flow of material, if this is without risk. Dike the spilled material, where this is
possible. Cover with plastic sheet to prevent spreading. Absorb in vermiculite, dry sand or earth and
place into containers. Following product recovery, flush area with water.
Small Spills: Wipe up with absorbent material (e.g. cloth, fleece). Clean surface thoroughly to
remove residual contamination.
Never return spills to original containers for re-use. For waste disposal, see section 13 of the SDS.
Environmental precautions Avoid discharge into drains, water courses or onto the ground.
SECTION 7. HANDLING AND STORAGE
Precautions for safe handling Observe good industrial hygiene practices.
Conditions for safe storage,
including any
incompatibilities
Store in original tightly closed container. Store away from incompatible materials (see Section 10 of
the SDS).
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Occupational exposure limits No exposure limits noted for ingredient(s).
Biological limit values No biological exposure limits noted for the ingredient(s).
Appropriate engineering
controls
Good general ventilation (typically 10 air changes per hour) should be used. Ventilation rates should
be matched to conditions. If applicable, use process enclosures, local exhaust ventilation, or other
engineering controls to maintain airborne levels below recommended exposure limits. If exposure
limits have not been established, maintain airborne levels to an acceptable level.
Individual protection measures, such as personal protective equipment
Eye/face protection Wear safety glasses with side shields (or goggles).
Skin protection
Hand protection Wear appropriate chemical resistant gloves.
Other Wear suitable protective clothing.
Respiratory protection In case of insufficient ventilation, wear suitable respiratory equipment.
Thermal hazards Wear appropriate thermal protective clothing, when necessary.
General hygiene
considerations
Always observe good personal hygiene measures, such as washing after handling the material and
before eating, drinking, and/or smoking. Routinely wash work clothing and protective equipment to
remove contaminants.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Appearance
Physical state Liquid.
Form Liquid.
Color Colorless.
Odor Odorless.
Odor threshold Not available.
pH Not available.
Melting point/freezing point 32 °F (0 °C) estimated
Initial boiling point and
boiling range
212 °F (100 °C) estimated
Flash point Not available.
Evaporation rate Not available.
Flammability (solid, gas) Not available.
Upper/lower flammability or explosive limits
Flammability limit - lower
(%)
Not available.
Flammability limit -
upper (%)
Not available.
Explosive limit - lower
(%)
Not available.
Explosive limit - upper
(%)
Not available.
Vapor pressure Not available.
Vapor density Not available.
Relative density Not available.
Solubility(ies)
Solubility (water) Not available.
Partition coefficient
(n-octanol/water)
Not available.
Auto-ignition temperature Not available.
Decomposition temperature Not available.
Viscosity Not available.
Other information
Density 1 g/cm3 estimated
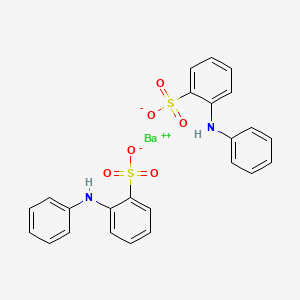
Molecular formula Ba(SO3C6H4NHC6H5)2
Percent volatile 99 % estimated
Specific gravity 1 estimated
SECTION 10. STABILITY AND REACTIVITY
Reactivity The product is stable and non-reactive under normal conditions of use, storage and transport.
Chemical stability Material is stable under normal conditions.
Possibility of hazardous
reactions
No dangerous reaction known under conditions of normal use.
Conditions to avoid Contact with incompatible materials.
Incompatible materials Strong oxidizing agents.
Hazardous decomposition
products
No hazardous decomposition products are known.
SECTION 11. TOXICOLOGICAL INFORMATION
Information on likely routes of exposure
Inhalation No adverse effects due to inhalation are expected.
Skin contact No adverse effects due to skin contact are expected.
Eye contact Direct contact with eyes may cause temporary irritation.
Ingestion Expected to be a low ingestion hazard.
Symptoms related to the
physical, chemical and
toxicological characteristics
Direct contact with eyes may cause temporary irritation.
Information on toxicological effects
Acute toxicity Not available.
Skin corrosion/irritation Prolonged skin contact may cause temporary irritation.
Serious eye damage/eye
irritation
Direct contact with eyes may cause temporary irritation.
Respiratory or skin sensitization
Respiratory sensitization Not available.
Skin sensitization This product is not expected to cause skin sensitization.
Germ cell mutagenicity No data available to indicate product or any components present at greater than 0.1% are
mutagenic or genotoxic.
Carcinogenicity This product is not considered to be a carcinogen by IARC, ACGIH, NTP, or OSHA.
Reproductive toxicity This product is not expected to cause reproductive or developmental effects.
Specific target organ toxicity
- single exposure
Not classified.
Specific target organ toxicity
- repeated exposure
Not classified.
Aspiration hazard Not available.
SECTION 12. ECOLOGICAL INFORMATION
Ecotoxicity The product is not classified as environmentally hazardous. However, this does not exclude the
possibility that large or frequent spills can have a harmful or damaging effect on the environment.
Persistence and degradability No data is available on the degradability of this product.
Bioaccumulative potential No data available.
Mobility in soil No data available.
Other adverse effects No other adverse environmental effects (e.g. ozone depletion, photochemical ozone creation
potential, endocrine disruption, global warming potential) are expected from this component.
SECTION 13. DISPOSAL CONSIDERATIONS
Disposal instructions Collect and reclaim or dispose in sealed containers at licensed waste disposal site.
Local disposal regulations Dispose in accordance with all applicable regulations.
Hazardous waste code The waste code should be assigned in discussion between the user, the producer and the waste
disposal company.
Waste from residues /
unused products
Dispose of in accordance with local regulations. Empty containers or liners may retain some product
residues. This material and its container must be disposed of in a safe manner (see: Disposal
instructions).
Contaminated packaging Empty containers should be taken to an approved waste handling site for recycling or disposal.
Since emptied containers may retain product residue, follow label warnings even after container is
emptied.
SECTION 14. TRANSPORT INFORMATION
DOT
Not regulated as dangerous goods.
IATA
Not regulated as dangerous goods.
IMDG
Not regulated as dangerous goods.
Transport in bulk according to Not established.
Annex II of MARPOL 73/78
and the IBC Code
SECTION 15. REGULATORY INFORMATION
US federal regulations One or more components are not listed on TSCA.
This product is not known to be a "Hazardous Chemical" as defined by the OSHA Hazard
Communication Standard, 29 CFR 1910.1200.
TSCA Section 12(b) Export Notification (40 CFR 707, Subpt. D)
Not regulated.
CERCLA Hazardous Substance List (40 CFR 302.4)
Not listed.
SARA 304 Emergency release notification
Not regulated.
Superfund Amendments and Reauthorization Act of 1986 (SARA)
Hazard categories Immediate Hazard - No
Delayed Hazard - No
Fire Hazard - No
Pressure Hazard - No
Reactivity Hazard - No
SARA 302 Extremely hazardous substance
Not listed.
SARA 311/312
Hazardous chemical
No
SARA 313 (TRI reporting)
Not regulated.
Other federal regulations
Clean Air Act (CAA) Section 112 Hazardous Air Pollutants (HAPs) List
Not regulated.
Clean Air Act (CAA) Section 112(r) Accidental Release Prevention (40 CFR 68.130)
Not regulated.
Safe Drinking Water Act
(SDWA)
Not regulated.
US state regulations
US. California Controlled Substances. CA Department of Justice (California Health and Safety Code Section 11100)
Not listed.
US. Massachusetts RTK - Substance List
Not regulated.
US. New Jersey Worker and Community Right-to-Know Act
Not listed.
US. Pennsylvania Worker and Community Right-to-Know Law
Not listed.
US. Rhode Island RTK
Not regulated.
US. California Proposition 65
California Safe Drinking Water and Toxic Enforcement Act of 1986 (Proposition 65): This material is not known to contain any
chemicals currently listed as carcinogens or reproductive toxins.
International Inventories
Country(s) or region Inventory name On inventory (yes/no)*
Australia Australian Inventory of Chemical Substances (AICS) No
Canada Domestic Substances List (DSL) No
Canada Non-Domestic Substances List (NDSL) No
China Inventory of Existing Chemical Substances in China (IECSC) No
Europe European Inventory of Existing Commercial Chemical Substances No
(EINECS)
Europe European List of Notified Chemical Substances (ELINCS) No
Japan Inventory of Existing and New Chemical Substances (ENCS) No
Korea Existing Chemicals List (ECL) No
New Zealand New Zealand Inventory No
Philippines Philippine Inventory of Chemicals and Chemical Substances No
(PICCS)
United States & Puerto Rico Toxic Substances Control Act (TSCA) Inventory No
*A "Yes" indicates that all components of this product comply with the inventory requirements administered by the governing country(s)
A "No" indicates that one or more components of the product are not listed or exempt from listing on the inventory administered by the governing
country(s).
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 Barium is a member of the alkaline-earth metals. The barium atom has a radius of 222 pm and a Van der Waals radius of 268 pm. Barium was discovered by Carl Wilhelm Scheele in 1772 and first isolated by Humphry Davy in 1808.
Barium is a member of the alkaline-earth metals. The barium atom has a radius of 222 pm and a Van der Waals radius of 268 pm. Barium was discovered by Carl Wilhelm Scheele in 1772 and first isolated by Humphry Davy in 1808.  In its elemental form, barium is a soft, silvery-gray metal. Industrial applications for barium include acting as a "getter," or unwanted gas remover, for vacuum tubes, and as an additive to steel and cast iron. Barium is also alloyed with silicon and aluminum in load-bearing alloys. The main commercial source of barium is the mineral barite (BaSO4); it does not occur naturally as a free element . The name barium is derived from the Greek word "barys," meaning heavy.
In its elemental form, barium is a soft, silvery-gray metal. Industrial applications for barium include acting as a "getter," or unwanted gas remover, for vacuum tubes, and as an additive to steel and cast iron. Barium is also alloyed with silicon and aluminum in load-bearing alloys. The main commercial source of barium is the mineral barite (BaSO4); it does not occur naturally as a free element . The name barium is derived from the Greek word "barys," meaning heavy.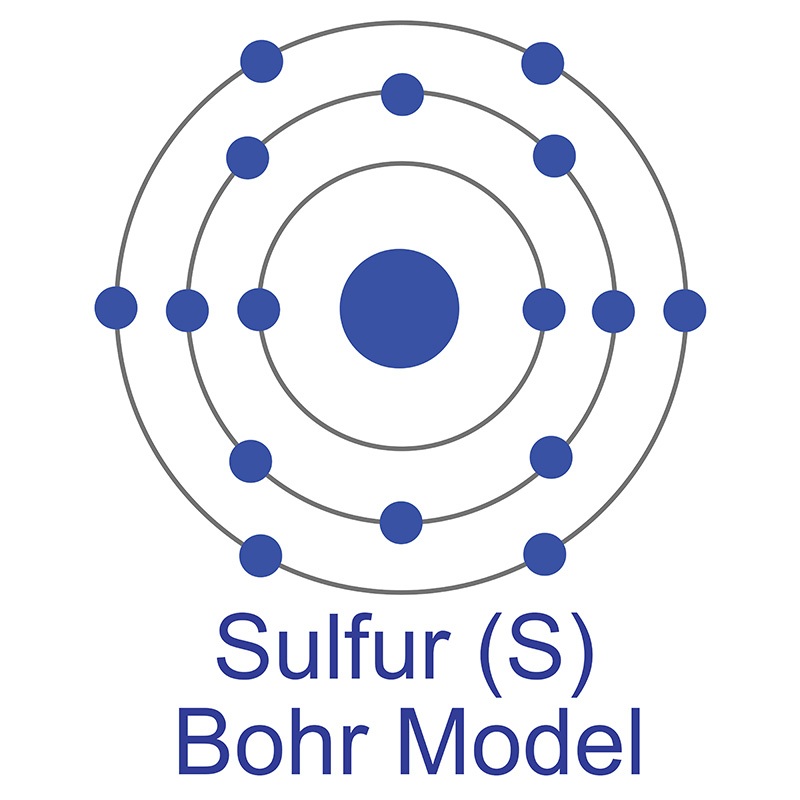 The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.
The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.