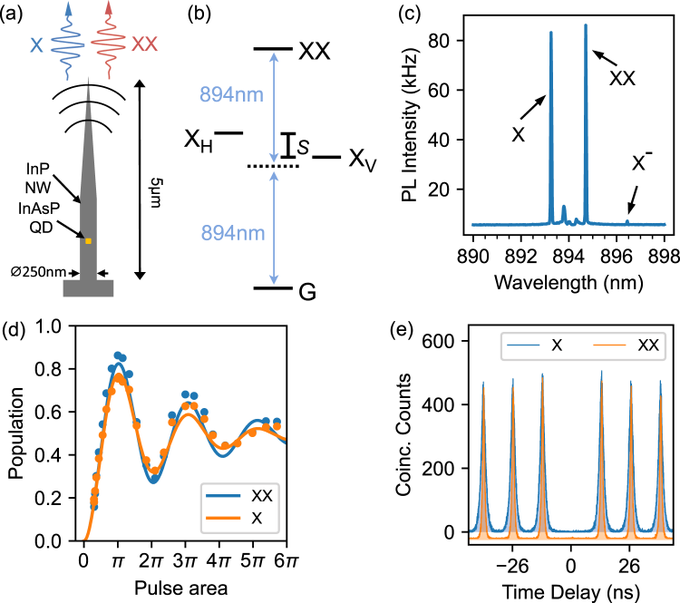
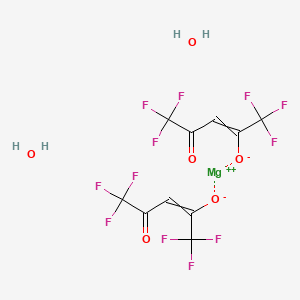
SECTION 1. IDENTIFICATION
Product Name: Magnesium Hexafluoroacetylacetonate Dihydrate
Product Number: All applicable American Elements product codes, e.g. MG-FACA-018-C.2HYD
CAS #: 19648-85-2
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
This product does not meet the criteria for classification in any hazard class according to Regulation (EC) No 1272/2008
Label elements
Pictograms or hazard symbols None
Signal word No signal word
Hazard statements None
Precautionary statements None
Other hazards Results of PBT and vPvB assessment
PBT: Not applicable
vPvB: Not applicable
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
Components: Bis(hexafluoroacetylacetonato)magnesium(II) Hydrate
CAS RN: 19648-85-2
Synonyms: Hexafluoroacetylacetono Magnesium(II) Salt Hydrate , Magnesium(II) Hexafluoroacetylacetonate
Hydrate
Chemical Formula: C10H2F12MgO4・xH2O
SECTION 4. FIRST AID MEASURES
Description of first aid measures
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing. Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with water/shower. If skin irritation
or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do.
Continue rinsing. If eye irritation persists: Get medical advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-tight goggles.
Most important symptoms and effects, both acute and delayed
No data available
Indication of any immediate medical attention and special treatment needed
No data available
SECTION 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media: Dry chemical, foam, water spray, carbon dioxide.
Special hazards arising from the substance or mixture
Take care as it may decompose upon combustion or in high temperatures to generate poisonous fume.
Carbon dioxide, Carbon monoxide, Hydrogen fluoride
Advice for firefighters Fire-extinguishing work is done from the windward and the suitable fire-extinguishing method according
to the surrounding situation is used. Uninvolved persons should evacuate to a safe place. In case of fire in the surroundings: Remove movable containers if safe to do so. When extinguishing fire, be sure to wear personal protective equipment
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Keep people away from and upwind of spill/leak. Entry to
non-involved personnel should be controlled around the leakage area by roping off, etc
Environmental precautions Prevent product from entering drains
Methods and materials for containment and cleaning up
Sweep dust to collect it into an airtight container, taking care not to disperse it. Adhered or collected
material should be promptly disposed of, in accordance with appropriate laws and regulations.
SECTION 7. HANDLING AND STORAGE
7.1 Precautions for safe handling Handling is performed in a well ventilated place. Wear suitable protective equipment. Prevent
dispersion of dust. Wash hands and face thoroughly after handling. Use a local exhaust if dust or
aerosol will be generated. Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any incompatibilities
Keep container tightly closed. Store in a cool and dark place. Store away from incompatible materials such as oxidizing agents.
Specific end use(s) No further relevant information available.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Control parameters No data available
Exposure controls Install a closed system or local exhaust as possible so that workers should not be exposed directly.
Also install safety shower and eye bath. Respiratory protection: Dust respirator. Follow local and national regulations. Hand protection: Protective gloves. Eye protection: Safety glasses. A face-shield, if the situation requires. Skin and body protection: Protective clothing. Protective boots, if the situation requires
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Form: Crystal - Powder
Colour: White - Slightly pale yellow red
Odour: No data available
pH: No data available
Melting point/freezing point: No data available
Boiling point/range: No data available
Flash point: No data available (Calculated value:>60°C)
Flammability or explosive limits: Lower: No data available Upper: No data available Relative density: No data available Solubility(ies): [Water] No data available [Other solvents] No data available Partition coefficient:
n-octanol/water:
No data available
Autoignition temperature: No data available
Decomposition temperature: No data available
Dynamic Viscosity:
No data available
Kinematic viscosity: No data available
Other safety information No data available
SECTION 10. STABILITY AND REACTIVITY
Reactivity No data available
Chemical stability Stable under proper conditions.
Possibility of hazardous reactions No special reactivity has been reported.
Conditions to avoid No data available
Incompatible materials Oxidizing agents
Hazardous decomposition products Carbon dioxide, Carbon monoxide, Hydrogen fluoride
SECTION 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute Toxicity:
Skin corrosion/irritation:
Serious eye damage/irritation:
Germ cell mutagenicity:
Carcinogenicity:
Reproductive toxicity:
SECTION 12. ECOLOGICAL INFORMATION
Toxicity Fish: No data available Crustacea: No data available Algae: No data available
Persistence and degradability No data available
Bioaccumulative potential No data available
Mobility in soil Log Pow: No data available Soil adsorption (Koc): No data available Henry's Law (PaM 3/mol): No data available
Results of PBT and vPvB assessment
PBT: Not applicable
vPvB: Not applicable
Other adverse effects No data available
SECTION 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material with a combustible solvent
and burn in a chemical incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
SECTION 14. TRANSPORT INFORMATION
UN number Not listed
UN proper shipping name
ADR/RID Not listed
IMDG/IMO Not listed
ICAO/IATA Not listed
Transport hazard class(es)
ADR/RID Does not correspond to the classification standard of the United Nations
IMDG/IMO Does not correspond to the classification standard of the United Nations
ICAO/IATA Does not correspond to the classification standard of the United Nations
Packaging group
ADR/RID -
IMDG/IMO -
ICAO/IATA -
Environmental hazards
Marine pollutant -
Special precautions for user No data available
SECTION 15. REGULATORY INFORMATION
Safety, health and environmental regulations/legislation specific for the substance or mixture
Substance of Very High Concern (SVHC) according to the
REACH Regulations (EC) No.1907/2006
Not listed
Chemical safety assessment A chemical safety assessment has not been carried out.
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 See more Magnesium products.
See more Magnesium products. In its elemental form, magnesium has a shiny grey metallic appearance and is an extremely reactive. It is can be found in minerals such as brucite, carnallite, dolomite, magnesite, olivine and talc. Commercially, magnesium is primarily used in the creation of strong and lightweight
In its elemental form, magnesium has a shiny grey metallic appearance and is an extremely reactive. It is can be found in minerals such as brucite, carnallite, dolomite, magnesite, olivine and talc. Commercially, magnesium is primarily used in the creation of strong and lightweight