About Moscovium
Moscovium (Mc), formerly known as Ununpentium (Uup), is a P-Block, Group 15, Period 7 element with atomic number 115. Synthesis of ununpentium was first reported in 2004 by a team of Russian scientists at the Joint Institute for Nuclear Research in Dubna, Russia and a team of American Scientists at the Lawrence Livermore National Laboratory in Livermore, California, USA, which were formally accepted by IUPAC in 2015. The collaboration synthesized the superheavy element by bombarding americium-243 with calcium-48 ions. The name of Moscovium refers to the Moscow Oblast, the location of the Joint Institute.
Though few properties have been observed of the highly unstable element, Moscovium is expected to share characteristics of the pnictogen group of elements (group 15, below bismuth) based on its position in the periodic table.
Moscovium Properties
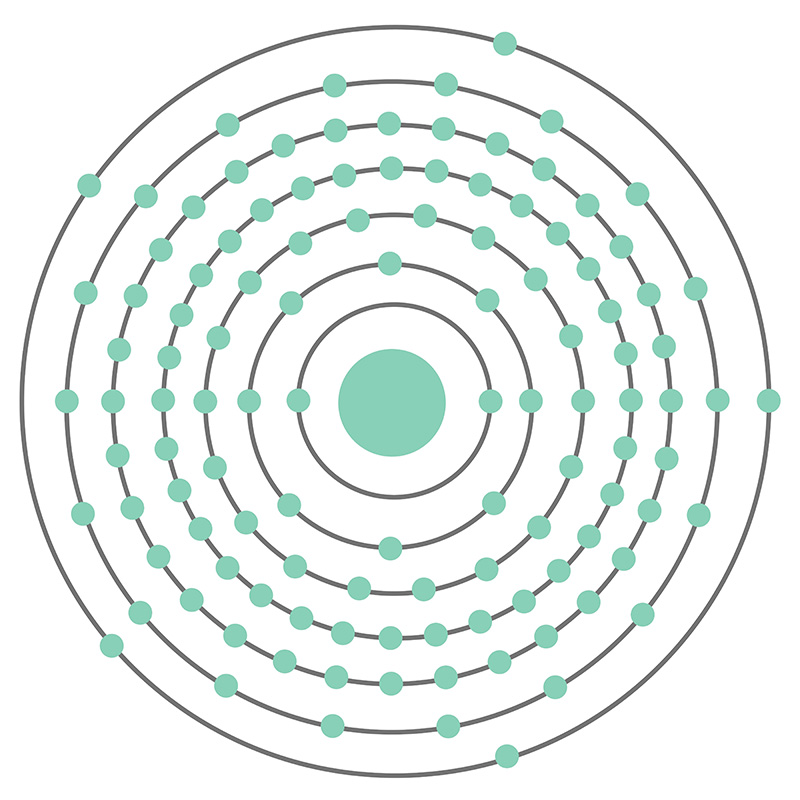
 Moscovium is a P-Block, Group 15, Period 7 element. The number of electrons in each of Ununpentium's shells is 2, 8, 18, 32, 32, 18, 5 and its electron configuration is [Rn] 5f14 6d10 7s27p3. In its elemental form Moscovium's CAS number is 54085-64-2. Moscovium was first reported in 2003 by a team of Russian scientists at the Joint Institute for Nuclear Research in Dubna, Russia and a team of American Scientists at the Lawrence Livermore National Laboratory in Livermore, California, USA. Little is known about the element, its appearance is unknown, and it has no known uses.
Moscovium is a P-Block, Group 15, Period 7 element. The number of electrons in each of Ununpentium's shells is 2, 8, 18, 32, 32, 18, 5 and its electron configuration is [Rn] 5f14 6d10 7s27p3. In its elemental form Moscovium's CAS number is 54085-64-2. Moscovium was first reported in 2003 by a team of Russian scientists at the Joint Institute for Nuclear Research in Dubna, Russia and a team of American Scientists at the Lawrence Livermore National Laboratory in Livermore, California, USA. Little is known about the element, its appearance is unknown, and it has no known uses.
Moscovium information, including properties, research, applications and other useful facts are discussed below. Scientific facts such as the atomic structure, ionization energy, abundance on Earth, conductivity and thermal properties are included.
| Symbol: |
Mc |
| Atomic Number: |
115 |
| Atomic Weight: |
288 |
| Element Category: |
unknown |
| Group, Period, Block: |
15 (pnictogens), 7, p |
| Color: |
unknown (presumably metallic/ silvery white/ gray) |
| Other Names: |
Ununpentium |
| Melting Point: |
-430 °C, -810 °F, -700 K |
| Boiling Point: |
-1100 °C, -2000 °F, -1400 K |
| Density: |
11 g·cm3 (predicted) |
| Liquid Density @ Melting Point: |
N/A |
| Density @ 20°C: |
N/A |
| Density of Solid: |
13000 (predicted) kg·m3 |
| Specific Heat: |
N/A |
| Superconductivity Temperature: |
N/A |
| Triple Point: |
N/A |
| Critical Point: |
N/A |
| Heat of Fusion (kJ·mol-1): |
N/A |
| Heat of Vaporization (kJ·mol-1): |
N/A |
| Heat of Atomization (kJ·mol-1): |
N/A |
| Thermal Conductivity: |
N/A |
| Thermal Expansion: |
N/A |
| Electrical Resistivity: |
N/A |
| Tensile Strength: |
N/A |
| Molar Heat Capacity: |
N/A |
| Young's Modulus: |
N/A |
| Shear Modulus: |
N/A |
| Bulk Modulus: |
N/A |
| Poisson Ratio: |
N/A |
| Mohs Hardness: |
N/A |
| Vickers Hardness: |
N/A |
| Brinell Hardness: |
N/A |
| Speed of Sound: |
N/A |
| Pauling Electronegativity: |
N/A |
| Sanderson Electronegativity: |
N/A |
| Allred Rochow Electronegativity: |
N/A |
| Mulliken-Jaffe Electronegativity: |
N/A |
| Allen Electronegativity: |
N/A |
| Pauling Electropositivity: |
N/A |
| Reflectivity (%): |
N/A |
| Refractive Index: |
N/A |
| Electrons: |
115 |
| Protons: |
115 |
| Neutrons: |
174 |
| Electron Configuration: |
[Rn] 5f14 6d10 7s27p3 |
| Atomic Radius: |
200 pm (predicted) |
Atomic Radius,
non-bonded (Å): |
Unknown |
| Covalent Radius: |
162 pm (predicted) |
| Covalent Radius (Å): |
1.62 |
| Van der Waals Radius: |
N/A |
| Oxidation States: |
1,3 (predicted) |
| Phase: |
Solid (predicted) |
| Crystal Structure: |
unknown |
| Magnetic Ordering: |
unknown |
| Electron Affinity (kJ·mol-1) |
Unknown |
| 1st Ionization Energy: |
538.4 kJ·mol-1 (estimated) |
| 2nd Ionization Energy: |
N/A |
| 3rd Ionization Energy: |
N |
| CAS Number: |
54085-64-2 |
| EC Number: |
N/A |
| MDL Number: |
N/A |
| Beilstein Number: |
N/A |
| SMILES Identifier: |
N/A |
| InChI Identifier: |
N/A |
| InChI Key: |
N/A |
| PubChem CID: |
N/A |
| ChemSpider ID: |
N/A |
| Earth - Total: |
N/A |
| Mercury - Total: |
N/A |
| Venus - Total: |
N/A |
| Earth - Seawater (Oceans), ppb by weight: |
N/A |
| Earth - Seawater (Oceans), ppb by atoms: |
N/A |
| Earth - Crust (Crustal Rocks), ppb by weight: |
N/A |
| Earth - Crust (Crustal Rocks), ppb by atoms: |
N/A |
| Sun - Total, ppb by weight: |
N/A |
| Sun - Total, ppb by atoms: |
N/A |
| Stream, ppb by weight: |
N/A |
| Stream, ppb by atoms: |
N/A |
| Meterorite (Carbonaceous), ppb by weight: |
N/A |
| Meterorite (Carbonaceous), ppb by atoms: |
N/A |
| Typical Human Body, ppb by weight: |
N/A |
| Typical Human Body, ppb by atom: |
N/A |
| Universe, ppb by weight: |
N/A |
| Universe, ppb by atom: |
N/A |
| Discovered By: |
Joint Institute for Nuclear Research and Lawrence Livermore National Laboratory |
| Discovery Date: |
2003 |
| First Isolation: |
N/A |
Moscovium Isotopes
Moscovium is an artificial element. Like all artificial elements, it has no stable isotopes.
| Nuclide |
Isotopic Mass |
Half-Life |
Mode of Decay |
Nuclear Spin |
Magnetic Moment |
Binding Energy (MeV) |
Natural Abundance
(% by atom) |
| 287Mc | 287.19070(52)# | 32(+155-14) ms | α to 283Nh | N/A | N/A | N/A | - |
| 288Mc | 288.19274(62)# | 87(+105-30) ms | α to 284Nh | N/A | N/A | N/A | - |
| 289Mc | 289.19363(89)# | 220 ms | α to 285Nh | N/A | N/A | N/A | - |
| 290Mc | 290.19598(73)# | 16 ms | α to 286Nh | N/A | N/A | N/A | - |

 Moscovium is a P-Block, Group 15, Period 7 element. The number of electrons in each of Ununpentium's shells is 2, 8, 18, 32, 32, 18, 5 and its electron configuration is [Rn] 5f14 6d10 7s27p3. In its elemental form Moscovium's CAS number is 54085-64-2. Moscovium was first reported in 2003 by a team of Russian scientists at the Joint Institute for Nuclear Research in Dubna, Russia and a team of American Scientists at the Lawrence Livermore National Laboratory in Livermore, California, USA. Little is known about the element, its appearance is unknown, and it has no known uses.
Moscovium is a P-Block, Group 15, Period 7 element. The number of electrons in each of Ununpentium's shells is 2, 8, 18, 32, 32, 18, 5 and its electron configuration is [Rn] 5f14 6d10 7s27p3. In its elemental form Moscovium's CAS number is 54085-64-2. Moscovium was first reported in 2003 by a team of Russian scientists at the Joint Institute for Nuclear Research in Dubna, Russia and a team of American Scientists at the Lawrence Livermore National Laboratory in Livermore, California, USA. Little is known about the element, its appearance is unknown, and it has no known uses.