SECTION 1. IDENTIFICATION
Product Name: Thulium Metal
Product Number: All applicable American Elements product codes, e.g. TM-M-02, TM-M-03, TM-M-04, TM-M-05
CAS #: 7440-30-4
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
1093 Broxton Ave. Suite 2000
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America +1 800-424-9300
International +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture in accordance with 29 CFR 1910 (OSHA HCS)
GHS02 Flame
Flam. Sol. 1 H228 Flammable solid.
Water-react. 1 H260 In contact with water releases flammable gases which may ignite spontaneously.
Hazards not otherwise classified
No information known.
Label elements
GHS label elements
The product is classified and labeled in accordance with 29 CFR 1910 (OSHA HCS)
Hazard pictograms

GHS02
Signal word
Danger
Hazard statements
H228 Flammable solid.
H260 In contact with water releases flammable gases which may ignite spontaneously.
Precautionary statements
P210 Keep away from heat/sparks/open flames/hot surfaces. No smoking.
P231+P232 Handle under inert gas. Protect from moisture.
P280 Wear protective gloves/protective clothing/eye protection/face protection.
P370+P378 In case of fire: Use for extinction: Special powder for metal fires.
P402+P404 Store in a dry place. Store in a closed container.
P501 Dispose of contents/container in accordance with local/regional/
national/international regulations.
WHMIS classification
B4 - Flammable solid
B6 - Reactive flammable material
Classification system
HMIS ratings (scale 0-4)
(Hazardous Materials Identification System)
HEALTH
FIRE
REACTIVITY
1
3
2
Health (acute effects) = 1
Flammability = 3
Physical Hazard = 2
Other hazards
Results of PBT and vPvB assessment
PBT:
Not applicable.
vPvB:
Not applicable
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Chemical characterization: Substances
CAS# Description:
7440-30-4 thulium
Identification number(s):
EC number:
231-140-2
SECTION 4. FIRST AID MEASURES
Description of first aid measures
After inhalation
Supply fresh air. If required, provide artificial respiration. Keep patient warm.
Seek immediate medical advice.
After skin contact
Immediately wash with water and soap and rinse thoroughly.
Seek immediate medical advice.
After eye contact
Rinse opened eye for several minutes under running water. Then consult a doctor.
After swallowing
Seek medical treatment.
Information for doctor
Most important symptoms and effects, both acute and delayed
No further relevant information available.
Indication of any immediate medical attention and special treatment needed
No further relevant information available.
SECTION 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing agents
Special powder for metal fires. Do not use water.
For safety reasons unsuitable extinguishing agents
Water
Special hazards arising from the substance or mixture
If this product is involved in a fire, the following can be released:
Metal oxide fume
Advice for firefighters
Protective equipment:
Wear self-contained respirator.
Wear fully protective impervious suit.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Wear protective equipment. Keep unprotected persons away.
Ensure adequate ventilation
Keep away from ignition sources
Environmental precautions:
Do not allow material to be released to the environment without proper governmental permits.
Methods and material for containment and cleaning up:
Keep away from ignition sources.
Ensure adequate ventilation.
Do not flush with water or aqueous cleansing agents
Prevention of secondary hazards:
Keep away from ignition sources.
Reference to other sections
See Section 7 for information on safe handling
See Section 8 for information on personal protection equipment.
See Section 13 for disposal information.
SECTION 7. HANDLING AND STORAGE
Handling
Precautions for safe handling
Handle under dry protective gas.
Keep container tightly sealed.
Store in cool, dry place in tightly closed containers.
Ensure good ventilation at the workplace.
Information about protection against explosions and fires:
Protect against electrostatic charges.
Conditions for safe storage, including any incompatibilities
Storage
Requirements to be met by storerooms and receptacles:
Store in a cool location.
Information about storage in one common storage facility:
Store away from air.
Store away from water/moisture.
Do not store together with acids.
Store away from oxidizing agents.
Store away from halogens.
Store away from acid chlorides.
Further information about storage conditions:
Store under dry inert gas.
This product is moisture sensitive.
This product is air sensitive.
Keep container tightly sealed.
Store in cool, dry conditions in well sealed containers.
Protect from humidity and water.
Specific end use(s)
No further relevant information available.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Additional information about design of technical systems:
Properly operating chemical fume hood designed for hazardous chemicals and having an average face velocity of at least 100 feet per minute.
Control parameters
Components with limit values that require monitoring at the workplace:
The product does not contain any relevant quantities of materials with critical values that have to be monitored at the workplace.
Additional information:
No data
Exposure controls
Personal protective equipment
General protective and hygienic measures
The usual precautionary measures for handling chemicals should be followed.
Keep away from foodstuffs, beverages and feed.
Remove all soiled and contaminated clothing immediately.
Wash hands before breaks and at the end of work.
Maintain an ergonomically appropriate working environment.
Breathing equipment:
Use suitable respirator when high concentrations are present.
Protection of hands:
Impervious gloves
Check protective gloves prior to each use for their proper condition.
The selection of suitable gloves not only depends on the material, but also on quality. Quality will vary from manufacturer to manufacturer.
Penetration time of glove material (in minutes)
Not determined
Eye protection:
Safety glasses
Full face protection
Body protection:
Protective work clothing.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
General Information
Appearance:
Form: Solid in various forms
Color: Grey to silver
Odor: Odorless
Odor threshold: Not determined.
pH-value: Not applicable.
Change in condition
Melting point/Melting range: 1545 °C (2813 °F)
Boiling point/Boiling range: 1725 °C (3137 °F)
Sublimation temperature / start: Not determined
Flammability (solid, gaseous)
Highly flammable.
Contact with water liberates extremely flammable gases.
Ignition temperature: Not determined
Decomposition temperature: Not determined
Auto igniting: Not determined.
Danger of explosion: Not determined.
Explosion limits:
Lower: Not determined
Upper: Not determined
Vapor pressure: Not applicable.
Density at 20 °C (68 °F): 9.32 g/cm³ (77.775 lbs/gal)
Relative density
Not determined.
Vapor density
Not applicable.
Evaporation rate
Not applicable.
Solubility in / Miscibility with Water: Contact with water releases flammable gases
Partition coefficient (n-octanol/water): Not determined.
Viscosity:
dynamic: Not applicable.
kinematic: Not applicable.
Other information
No further relevant information available.
SECTION 10. STABILITY AND REACTIVITY
Reactivity
In contact with water releases flammable gases which may ignite spontaneously.
Chemical stability
Stable under recommended storage conditions.
Thermal decomposition / conditions to be avoided:
Decomposition will not occur if used and stored according to specifications.
Possibility of hazardous reactions
Reacts with strong oxidizing agents
Contact with water releases flammable gases
Conditions to avoid
No further relevant information available.
Incompatible materials:
Acids
Oxidizing agents
Air
Halogens
Acid chlorides
Water/moisture
Hazardous decomposition products:
Metal oxide fume
SECTION 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity:
No effects known.
LD/LC50 values that are relevant for classification:
No data
Skin irritation or corrosion:
May cause irritation
Eye irritation or corrosion:
May cause irritation
Sensitization:
No sensitizing effects known.
Germ cell mutagenicity:
No effects known.
Carcinogenicity:
No classification data on carcinogenic properties of this material is available from the EPA, IARC, NTP, OSHA or ACGIH.
Reproductive toxicity:
No effects known.
Specific target organ system toxicity - repeated exposure:
No effects known.
Specific target organ system toxicity - single exposure:
No effects known.
Aspiration hazard:
No effects known.
Subacute to chronic toxicity:
No effects known.
Additional toxicological information:
To the best of our knowledge the acute and chronic toxicity of this substance is not fully known.
SECTION 12. ECOLOGICAL INFORMATION
Toxicity
Aquatic toxicity:
No further relevant information available.
Persistence and degradability
No further relevant information available.
Bioaccumulative potential
No further relevant information available.
Mobility in soil
No further relevant information available.
Additional ecological information:
General notes:
Do not allow material to be released to the environment without proper governmental permits.
Do not allow undiluted product or large quantities to reach ground water, water course or sewage system.
Avoid transfer into the environment.
Results of PBT and vPvB assessment
PBT:
Not applicable.
vPvB:
Not applicable.
Other adverse effects
No further relevant information available.
SECTION 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Recommendation
Consult state, local or national regulations to ensure proper disposal.
Uncleaned packagings:
Recommendation:
Disposal must be made according to official regulations.
SECTION 14. TRANSPORT INFORMATION
UN-Number
DOT, IMDG, IATA
UN3178
UN proper shipping name
DOT
Flammable solid, inorganic, n.o.s.
IMDG, IATA
FLAMMABLE SOLID, INORGANIC, N.O.S. (thulium)
Transport hazard class(es)
DOT
Class
4.1 Flammable solids, self-reactive substances and solid desensitised explosives.
Label
4.1
Class
4.1 (F3) Flammable solids, self-reactive substances and solid desensitised explosives
Label
4.1
IMDG, IATA
Class
4.1 Flammable solids, self-reactive substances and solid desensitised explosives.
Label
4.1
Packing group
DOT, IMDG, IATA
II
Environmental hazards:
Not applicable.
Special precautions for user
Warning: Flammable solids, self-reactive substances and solid desensitised explosives
Transport in bulk according to Annex II of MARPOL73/78 and the IBC Code
Not applicable.
Transport/Additional information:
DOT
Marine Pollutant (DOT):
No
UN "Model Regulation":
UN3178, Flammable solid, inorganic, n.o.s., 4.1, II
SECTION 15. REGULATORY INFORMATION
Safety, health and environmental regulations/legislation specific for the substance or mixture
GHS label elements
The product is classified and labeled in accordance with 29 CFR 1910 (OSHA HCS)
Hazard pictograms
GHS02
Signal word
Danger
Hazard statements
H228 Flammable solid.
H260 In contact with water releases flammable gases which may ignite spontaneously.
Precautionary statements
P210 Keep away from heat/sparks/open flames/hot surfaces. No smoking.
P231+P232 Handle under inert gas. Protect from moisture.
P280 Wear protective gloves/protective clothing/eye protection/face protection.
P370+P378 In case of fire: Use for extinction: Special powder for metal fires.
P402+P404 Store in a dry place. Store in a closed container.
P501 Dispose of contents/container in accordance with local/regional/
national/international regulations.
National regulations
All components of this product are listed in the U.S. Environmental Protection Agency Toxic Substances Control Act Chemical substance Inventory.
All components of this product are listed on the Canadian Domestic Substances List (DSL).
SARA Section 313 (specific toxic chemical listings)
Substance is not listed.
California Proposition 65
Prop 65 - Chemicals known to cause cancer
Substance is not listed.
Prop 65 - Developmental toxicity
Substance is not listed.
Prop 65 - Developmental toxicity, female
Substance is not listed.
Prop 65 - Developmental toxicity, male
Substance is not listed.
Information about limitation of use:
For use only by technically qualified individuals.
Other regulations, limitations and prohibitive regulations
Substance of Very High Concern (SVHC) according to the REACH Regulations (EC) No. 1907/2006.
Substance is not listed.
The conditions of restrictions according to Article 67 and Annex XVII of the Regulation (EC) No 1907/2006 (REACH) for the manufacturing, placing on the market and use must be observed.
Substance is not listed.
Annex XIV of the REACH Regulations (requiring Authorisation for use)
Substance is not listed.
Chemical safety assessment:
A Chemical Safety Assessment has not been carried out.
16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2016 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 Thulium emits blue light upon excitation, a property that is exploited in flat panel display screens. Thulium also has useful applications in low-levelradiation detection and other luminescence applications, such as halide discharge lamps. Flat panel screens depend critically on bright blue
Thulium emits blue light upon excitation, a property that is exploited in flat panel display screens. Thulium also has useful applications in low-levelradiation detection and other luminescence applications, such as halide discharge lamps. Flat panel screens depend critically on bright blue  emitters. Thulium is available as metal and compounds with purities from 99% to 99.999% (ACS grade to ultra-high purity). Elemental or metallic forms include pellets, rod, wire and granules for evaporation source material purposes.
emitters. Thulium is available as metal and compounds with purities from 99% to 99.999% (ACS grade to ultra-high purity). Elemental or metallic forms include pellets, rod, wire and granules for evaporation source material purposes.  Thulium oxide is available in powder and dense pellet form for such uses as optical coating and thin film applications. Oxides tend to be insoluble. Terbium fluorides is another insoluble form for uses in which oxygen is undesirable such as metallurgy, chemical and physical vapor deposition and in some optical coatings. Thulium is also available in soluble forms including chlorides, nitrates and acetates. These compounds can be manufactured as solutions at specified stoichiometries.
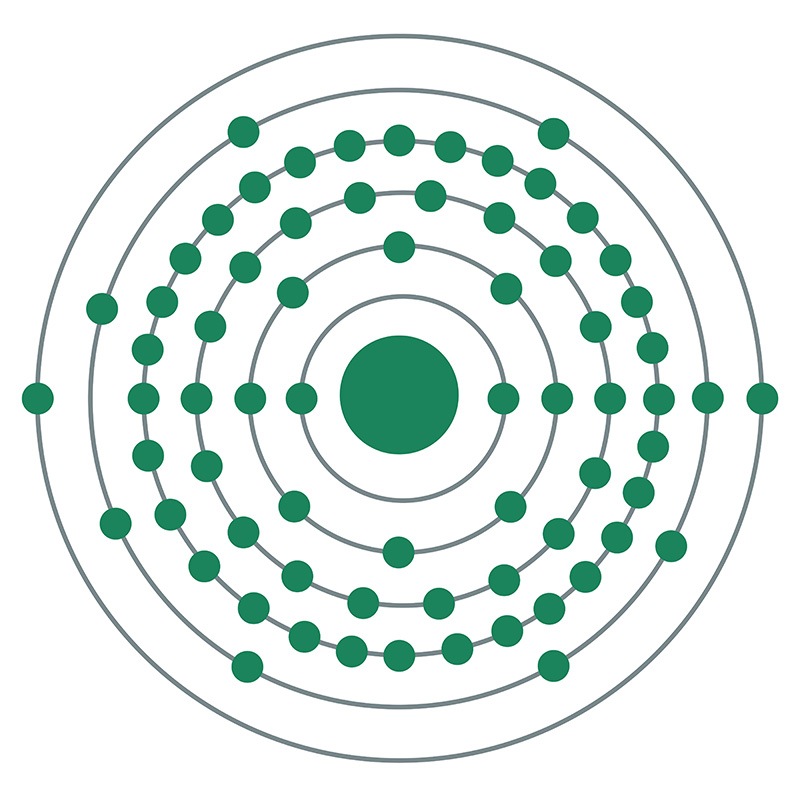
Thulium oxide is available in powder and dense pellet form for such uses as optical coating and thin film applications. Oxides tend to be insoluble. Terbium fluorides is another insoluble form for uses in which oxygen is undesirable such as metallurgy, chemical and physical vapor deposition and in some optical coatings. Thulium is also available in soluble forms including chlorides, nitrates and acetates. These compounds can be manufactured as solutions at specified stoichiometries.![]() Thulium is a Block F, Group 3, Period 6 element.
Thulium is a Block F, Group 3, Period 6 element.  , which is the ancient name of Scandinavia.
, which is the ancient name of Scandinavia.