SECTION 1. IDENTIFICATION
Product Name: Phosphorus Pentasulfide
Product Number: All applicable American Elements product codes, e.g. P-S-02
, P-S-03
, P-S-04
, P-S-05
CAS #: 1314-80-3
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
GHS Classification in accordance with 29 CFR 1910 (OSHA HCS)
Flammable solids (Category 1), H228
Chemicals which, in contact with water, emit flammable gases (Category 1), H260
Acute toxicity, Oral (Category 4), H302
Acute toxicity, Inhalation (Category 4), H332
Short-term (acute) aquatic hazard (Category 1), H400
Signal word
Danger
Hazard statement(s)
H228 Flammable solid.
H260 In contact with water releases flammable gases which may ignite spontaneously.
H302 + H332 Harmful if swallowed or if inhaled.
H400 Very toxic to aquatic life.
Precautionary statement(s)
P210 Keep away from heat/ sparks/ open flames/ hot surfaces. No smoking.
P223 Do not allow contact with water.
P231 + P232 Handle under inert gas. Protect from moisture.
P240 Ground/bond container and receiving equipment.
P241 Use explosion-proof electrical/ ventilating/ lighting/ equipment.
P261 Avoid breathing dust/ fume/ gas/ mist/ vapors/ spray.
P264 Wash skin thoroughly after handling.
P270 Do not eat, drink or smoke when using this product.
P271 Use only outdoors or in a well-ventilated area.
P273 Avoid release to the environment.
P280 Wear protective gloves/ eye protection/ face protection.
P301 + P312 + P330 IF SWALLOWED: Call a POISON CENTER/ doctor if you feel unwell. Rinse mouth.
P304 + P340 + P312 IF INHALED: Remove person to fresh air and keep comfortable for breathing. Call a POISON CENTER/ doctor if you feel unwell.
P335 + P334 Brush off loose particles from skin. Immerse in cool water/ wrap in wet bandages.
P370 + P378 In case of fire: Use dry sand, dry chemical or alcohol-resistant foam to extinguish.
P391 Collect spillage.
P402 + P404 Store in a dry place. Store in a closed container.
P501 Dispose of contents/ container to an approved waste disposal plant.
Hazards not otherwise classified (HNOC) or not covered by GHS
Contact with water liberates toxic gas.
Lachrymator., Stench.
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
Synonyms : Phosphorus(V) sulfide
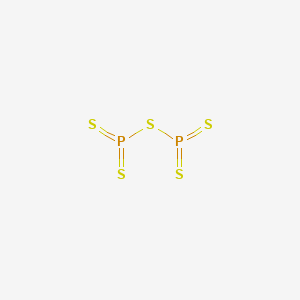
Formula : P2S5
Molecular weight : 222.27 g/mol
CAS-No. : 1314-80-3
SECTION 4. FIRST AID MEASURES
Description of first-aid measures
General advice
Show this material safety data sheet to the doctor in attendance.
If inhaled
After inhalation: fresh air. If breathing stops: mouth-to-mouth breathing or artificial
respiration. Oxygen if necessary. Immediately call in physician.
In case of skin contact
In case of skin contact: Take off immediately all contaminated clothing. Rinse skin with
water/ shower.
In case of eye contact
After eye contact: rinse out with plenty of water. Remove contact lenses.
If swallowed
After swallowing: immediately make victim drink water (two glasses at most). Consult a
physician.
Most important symptoms and effects, both acute and delayed
The most important known symptoms and effects are described in the labelling (see section
2.2) and/or in section 11
Indication of any immediate medical attention and special treatment needed
No data available
SECTION 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media
Carbon dioxide (CO2) Sand Dry powder Cement
Unsuitable extinguishing media
Foam Water
Special hazards arising from the substance or mixture
Sulfur oxides
Oxides of phosphorus
Not combustible.
May not get in touch with: Water
Ambient fire may liberate hazardous vapours.
Advice for firefighters
In the event of fire, wear self-contained breathing apparatus.
Further information
Suppress (knock down) gases/vapors/mists with a water spray jet. Prevent fire
extinguishing water from contaminating surface water or the ground water system.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Advice for non-emergency personnel: Avoid inhalation of dusts. Avoid substance contact.
Ensure adequate ventilation. Keep away from heat and sources of ignition. Evacuate the
danger area, observe emergency procedures, consult an expert.
For personal protection see section 8.
Environmental precautions
Do not let product enter drains. Risk of explosion.
Methods and materials for containment and cleaning up
Cover drains. Collect, bind, and pump off spills. Observe possible material restrictions
(see sections 7 and 10). Take up dry. Dispose of properly. Clean up affected area. Avoid
generation of dusts.
Reference to other sections
For disposal see section 13.
SECTION 7. HANDLING AND STORAGE
Precautions for safe handling
Advice on safe handling
Work under hood. Do not inhale substance/mixture. Keep workplace dry. Do not allow
product to come into contact with water.
Advice on protection against fire and explosion
Keep away from open flames, hot surfaces and sources of ignition.Take precautionary
measures against static discharge.
Hygiene measures
Change contaminated clothing. Preventive skin protection recommended. Wash hands after
working with substance.
For precautions see section 2.2.
Conditions for safe storage, including any incompatibilities
Storage conditions
Tightly closed. Keep away from heat and sources of ignition.
Never allow product to get in contact with water during storage.
Storage class (TRGS 510): 4.3: Hazardous materials, which set free flammable gases upon
contact with water
Specific end use(s)
Apart from the uses mentioned in section 1.2 no other specific uses are stipulated
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Exposure controls
Appropriate engineering controls
Change contaminated clothing. Preventive skin protection recommended. Wash hands
after working with substance.
Personal protective equipment
Eye/face protection
Use equipment for eye protection tested and approved under appropriate
government standards such as NIOSH (US) or EN 166(EU). Safety glasses
Skin protection
This recommendation applies only to the product stated in the safety data sheet,
supplied by us and for the designated use.
Body Protection
Flame retardant antistatic protective clothing.
Respiratory protection
required when dusts are generated.
Our recommendations on filtering respiratory protection are based on the following
standards: DIN EN 143, DIN 14387 and other accompanying standards relating to
the used respiratory protection system.
Control of environmental exposure
Do not let product enter drains. Risk of explosion.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Appearance Form: powder
Color: yellow
Odor Stench.
Odor Threshold No data available
pH No data available
Melting point/freezing point
Melting point/range: 280 - 284 °C (536 - 543 °F) - lit.
Initial boiling point and boiling range 514 °C 957 °F at 1,013 hPa
Flash point ()Not applicable
Evaporation rate No data available
Flammability (solid, gas) The substance or mixture is a flammable solid with the category 1.
Upper/lower flammability or explosive limits No data available
Vapor pressure 1 hPa at 300 °C (572 °F)
Vapor density No data available
Density 2.09 g/mL at 25 °C (77 °F) - lit.
Relative density No data available
Water solubility No data available
Partition coefficient: n-octanol/water No data available
Autoignition temperature No data available
Decomposition temperature No data available
Viscosity No data available
Explosive properties No data available
Oxidizing properties No data available
SECTION 10. STABILITY AND REACTIVITY
Reactivity
No data available
Chemical stability
sensitive to moisture
Possibility of hazardous reactions
Risk of explosion with:
Water
Oxidizing agents
Alcohols
Conditions to avoid
Do not allow water to enter container because of violent reaction.
Moisture.
Incompatible materials
No data available
SECTION 11. TOXICOLOGICAL INFORMATION
Acute toxicity
LD50 Oral - Rat - 389 mg/kg
LC50 Inhalation - 4 h - 1.5 mg/l
LD50 Dermal - Rabbit - 3,160 mg/kg
Remarks: Prolonged skin contact may cause skin irritation and/or dermatitis.
No data available
Skin corrosion/irritation
No data available
Serious eye damage/eye irritation
No data available
Respiratory or skin sensitization
No data available
Germ cell mutagenicity
No data available
Carcinogenicity
IARC: No ingredient of this product present at levels greater than or equal to 0.1% is
identified as probable, possible or confirmed human carcinogen by IARC.
NTP: No ingredient of this product present at levels greater than or equal to 0.1% is
identified as a known or anticipated carcinogen by NTP.
OSHA: No component of this product present at levels greater than or equal to 0.1% is
on OSHA’s list of regulated carcinogens.
Reproductive toxicity
No data available
No data available
Specific target organ toxicity - single exposure
No data available
Specific target organ toxicity - repeated exposure
No data available
Aspiration hazard
No data available
Additional Information
RTECS: TH4375000
Cough, Shortness of breath, Headache, Nausea, Vomiting, Pulmonary edema. Effects may
be delayed., Hydrogen sulfide is strongly bound to methemoglobin in a manner similar to
cyanide. Toxicologically, its reaction with enzymes in the blood stream inhibits cell
respiration resulting in pulmonary paralysis, sudden collapse, and death. It is recognized by
its characteristic odor of "rotten eggs". The detectable, minimum perceptible odor occurs at
0.13ppm, rapid olfactory fatigue can occur at high concentrations (>100 ppm). At
concentrations of 20ppm hydrogen sulfide begins acting as an irritant on the mucous
membranes of the eyes and respiratory tract and increases with concentration and
exposure time. Eye irritation is characterized by irritation of the conjunctiva with
photophobia to keratoconjunctivitis and vesiculation of the cornea epithelium. Prolonged
exposure to moderate concentrations (250ppm) may cause pulmonary edema. At
concentrations over 500ppm, drowsiness, dizziness, excitement, headache, unstable gait,
and other systemic symptoms occur within a few minutes. Sudden loss of consciousness
without premonition, anxiety, or sense of struggle are characteristic of acute exposure at
concentrations above 700ppm. At concentrations of 1000-2000ppm hydrogen sulfide is
rapidly absorbed through the lung into the blood. In this range a single inhalation may
cause coma and may be rapidly fatal. Initially hyperpnea occurs, followed by rapid collapse
and respiratory inhibition. At higher concentrations, hydrogen sulfide exerts an immediate
paralyzing effect on the respiratory centers. When concentration reaches 5000ppm,
imminent death almost always results.
Stomach - Irregularities - Based on Human Evidence
Stomach - Irregularities - Based on Human Evidence
SECTION 12. ECOLOGICAL INFORMATION
Toxicity
No data available
Persistence and degradability
Biodegradability Result: - Readily biodegradable.
Bioaccumulative potential
No data available
Mobility in soil
No data available
Results of PBT and vPvB assessment
PBT/vPvB assessment not available as chemical safety assessment not required/not
conducted
Other adverse effects
No data available
SECTION 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Product
Waste material must be disposed of in accordance with the national and local regulations.
Leave chemicals in original containers. No mixing with other waste. Handle uncleaned
containers like the product itself. See www.retrologistik.com for processes regarding the
return of chemicals and containers, or contact us there if you have further questions.
SECTION 14. TRANSPORT INFORMATION
DOT (US)
UN number: 1340 Class: 4.3 (4.1) Packing group: II
Proper shipping name: Phosphorus pentasulfide
Reportable Quantity (RQ): 100 lbs
Poison Inhalation Hazard: No
IMDG
UN number: 1340 Class: 4.3 (4.1) Packing group: II EMS-No: F-G, S-N
Proper shipping name: PHOSPHORUS PENTASULPHIDE
Marine pollutant : yes
IATA
UN number: 1340 Class: 4.3 (4.1) Packing group: II
Proper shipping name: Phosphorus pentasulphide
SECTION 15. REGULATORY INFORMATION
SARA 302 Components
This material does not contain any components with a section 302 EHS TPQ.
SARA 313 Components
This material does not contain any chemical components with known CAS numbers that
exceed the threshold (De Minimis) reporting levels established by SARA Title III, Section
313.
SARA 311/312 Hazards
Reactivity Hazard, Acute Health Hazard, Chronic Health Hazard
Massachusetts Right To Know Components
No components are subject to the Massachusetts Right to Know Act.
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 See more Phosphorus products.
See more Phosphorus products.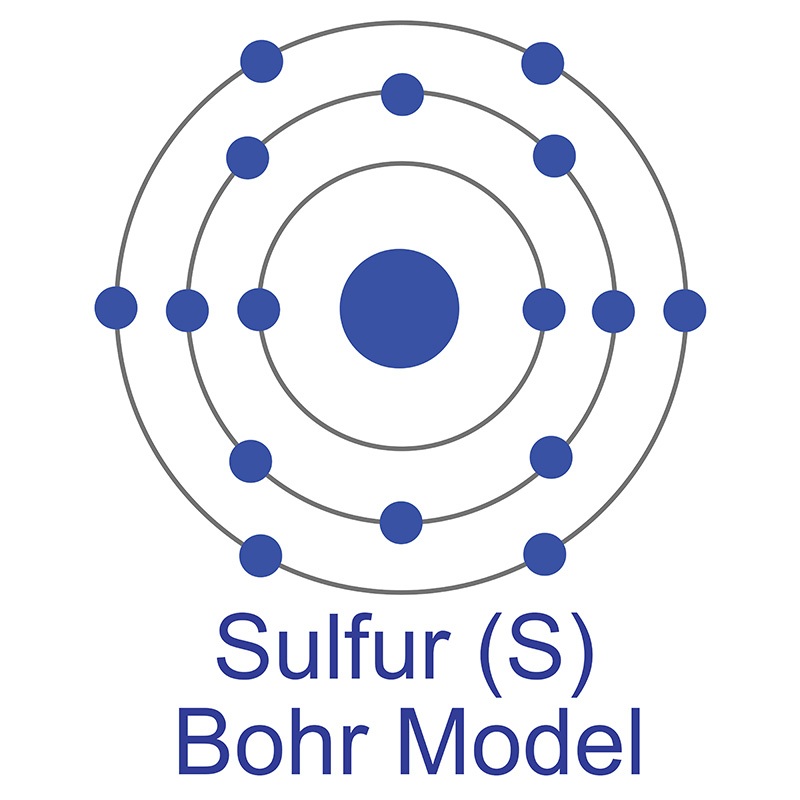 The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.
The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.