Bioceramics
Hydroxyapatite is a durable bioceramic similar to the naturally occurring mineral component found in bone and teeth. The compound is a calcium phosphate ceramic and is classified as bioactive in its function to support bone ingrowth and osseo-integration in orthopedic, dental and craniofacial applications.
Tricalcium phosphate ceramics are being tested for use as synthetic bone substitutes. The resorbable property of calcium phosphate is ideal in acting as temporary bone filler which dissolves over time to be replaced by natural bone.
Oxides
Zirconium oxide has a high melting point, strength and thermal stability making it useful in dentistry applications such as manufacturing corona and bridge frames, tooth root studs, and metal free dental implants.
Aluminum oxide or alumina has been implemented in dental restoration procedures including coating the outside surface of dental porcelain crowns.
Recent studies have found that magnesium oxide particle exhibit desirable properties for orthopedic tissue engineering. Additionally findings report improved fibroblast and osteoblast cellular attachment and subsequent growth on poly lactic acid in the presence of magnesium oxide particles.
Bioactive silica glass-ceramics bond to bone and are finding use in orthopedic applications. Researchers have found that mesoporous silica films serve as coatings on medical prostheses and implants. Another recent study has reported the use of sol-gel silica biomaterials for use bone tissue regeneration and in drug delivery systems.
Metals and Alloys
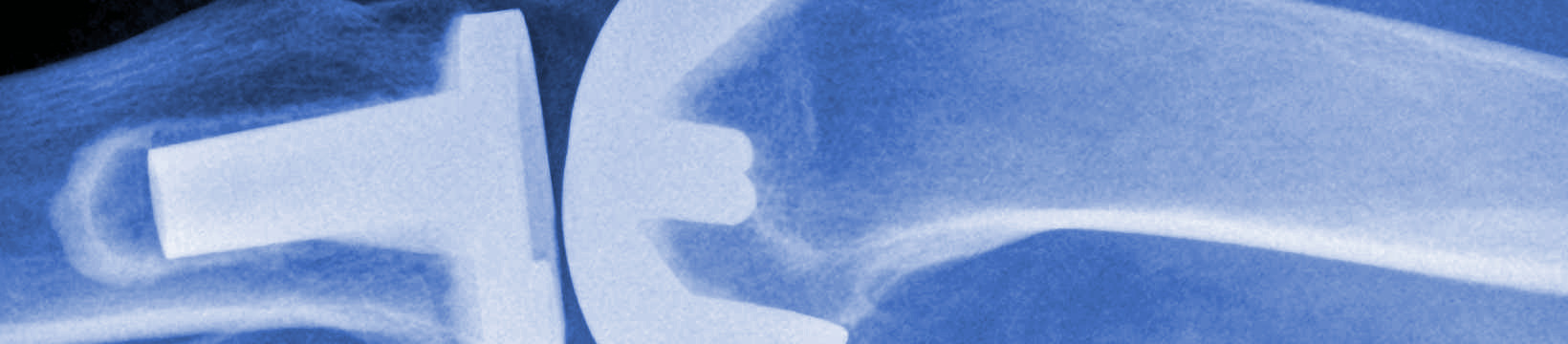
 Titanium metal is biocompatible and shows high corrosion resistance. Due to these beneficial properties, titanium is used in total joint replacement, dental implants, plates and screws. Metallic debris and higher risks of infection are often seen in tissue surrounding titanium implants. A variety of coatings and additives are being studied to reduce infection associated with titanium implants. Recent studies show its utility in bone implants as a powder in conjunction with hydroxyapatite and other compounds for bone restoration procedures.
Titanium metal is biocompatible and shows high corrosion resistance. Due to these beneficial properties, titanium is used in total joint replacement, dental implants, plates and screws. Metallic debris and higher risks of infection are often seen in tissue surrounding titanium implants. A variety of coatings and additives are being studied to reduce infection associated with titanium implants. Recent studies show its utility in bone implants as a powder in conjunction with hydroxyapatite and other compounds for bone restoration procedures.
Titanium alloys are often used in joint and bone implants. Their durability and flexibility make them ideal for implants such as stents. Due to their biomechanical compatibility titanium alloys are often used for femoral stems in hip prostheses. A common titanium alloy used in implants is titanium aluminum vanadium alloy (Ti6Al4V). The coating of titanium alloys is an active area of study due to surface wearing and friction factors. Titanium nitride is one such coating that has been approved by the Food and Drug Administration (FDA).
Silver alloys such as silver-gold, silver-copper, and silver-tin are routinely used in dental amalgams to fill cavities formed by tooth decay or broken teeth. Dental amalgam is often a self-hardening mixture of silver-tin-copper alloy powder and liquid mercury.
Cobalt alloys provide high strength properties and are used in dental and orthopedic applications. For example, cobalt-chromium-molybdenum (CoCrMo) alloy is commonly used for total joint arthroplasty. Other cobalt-based alloys such as cobalt-nickel-chrome-molybdenum alloy and wrought cobalt-chromium-tungsten-nickel-alloy are employed as materials in prosthetic and orthotic surgical implants.
Stainless steels used in biomedical applications are known as surgical stainless steel with reference to specific grades of stainless steel that display high levels of corrosion resistance. All steels are alloys of iron, carbon, and other elements; surgical stainless steel in particular contains chromium, nickel and molybdenum, which together lend characteristics such as high resistance to oxidation and surface scratches (from chromium), an even and polished finish (from nickel), and increased hardness with the ability to sustain a cutting edge (from molybdenum). Surgical steel is also easy to sterilize, making it an ideal material for medical instruments. Surgical implants and equipment that experience mechanical pressure such as bone fixation screws and prostheses are other examples of applications for stainless steels.
Magnesium alloys such as magnesium aluminum zinc are commonly used in implanted screws and plates that degrade in vivo.
Platinum and its alloys are stable at ambient temperatures and display resistance to corrosion and oxidation even when heated. Therefore, the metal and its alloys are used in a variety of surgical instruments.
Gold is resistant to corrosion and tarnishing and its alloys are commonly used in restorative dentistry, and more advanced medical applications.
Cermets
Ceramic and metallic composite (cermet) materials are utilized in applications requiring high temperature resistance and hardness in combination with ductility and compressibility. These properties in addition to biocompatibility make cermets particularly useful for surgical implants including bone implants and hip replacements components. Alumina, zirconia, calcium phosphate and glass ceramics are commonly found in biomaterials used for components of replacement joints.
Nanobiomaterials
Advancements in biomedical research have enabled the use of nanoparticles and material compounds comprised of gold, silver, and magnetic iron oxide for numerous applications such as targeted delivery of pharmaceuticals to tissues.
Magnetic nanoparticles are being studied for their applications in medical treatments and diagnostics such as cancer treatment and medical imaging. In one application, ferrous nanoparticles placed in a magnetic field generate significant heat, thereby killing cancer cells by localized hyperthermia. Additionally magnetic nanoparticles are being used in anesthetic procedures and other drug delivery procedures.
Gold nanoparticles or colloidal gold have been the focus of therapeutics research involving the use of gold compounds for gene-regulation, drug delivery structures, imaging agents and photoresponsive therapeutics.
Silver nanoparticles are commonly used to coat surfaces requiring antimicrobial resistance.
Surface functionalized nanomaterials such as drug coated-polymers, powders, and other porous materials are being studied for their applications in injectable or ingestible pharmaceuticals.
Ruthenium Metal Complexes
Ruthenium is a rare transition metal which forms metal organic complexes that have been studied for its applications as a fluorescent probe in biological imaging. Ruthenium-rhodamine is one example of a metal complex used as a fluorescent probe. Ruthenium complexes are also used as stains in electron microscopy studies. For example, ruthenium red is used as a tissue stain in histology research such as identifying amyloid plaques in Alzheimer’s disease. Ruthenium complexes are also being studied for their anti-cancer properties. In particular these complexes appear to be more aggressive against cancers for which platinum based compounds have been ineffective. In addition, ruthenium compounds can be transported by proteins containing nitrogen and sulfur. For example, ruthenium complexes can be transported to transferring receptors by binding to transferrin, making them ideal for targeted drug delivery to cancer cells expressing high levels of these receptors.