SECTION 1. IDENTIFICATION
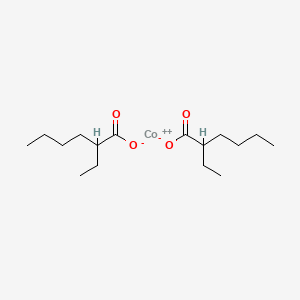
Product Name: Cobalt 2-Ethylhexanoate
Product Number: All applicable American Elements product codes, e.g. CO-2EH-02-LIQ
, CO-2EH-03-LIQ
, CO-2EH-04-LIQ
, CO-2EH-05-LIQ
CAS #: 136-52-7
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
GHS02 Flame
Flam. Liq. 3 H226 Flammable liquid and vapor.
GHS06 Skull and crossbones
Acute Tox. 3 H301 Toxic if swallowed.
GHS08 Health hazard
Carc. 2 H351 Suspected of causing cancer.
GHS label elements The product is classified and labeled according to the Globally Harmonized System (GHS).
Hazard pictograms
GHS02 GHS06 GHS08
Signal word Danger
Hazard-determining components of labeling:
cobalt bis(2-ethylhexanoate)
Hazard statements
H226 Flammable liquid and vapor.
H301 Toxic if swallowed.
H351 Suspected of causing cancer.
Precautionary statements
P101 If medical advice is needed, have product container or label at hand.
P102 Keep out of reach of children.
P103 Read label before use.
P210 Keep away from heat/sparks/open flames/hot surfaces. - No smoking.
P280 Wear protective gloves/protective clothing/eye protection/face protection.
P301+P310 IF SWALLOWED: Immediately call a POISON CENTER/ doctor.
P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if
present and easy to do. Continue rinsing.
P403+P233 Store in a well-ventilated place. Keep container tightly closed.
P501 Dispose of contents/container in accordance with local/regional/national/international
regulations.
Classification system:
NFPA ratings (scale 0 - 4)
2
2
0
Health = 2
Fire = 2
Reactivity = 0
HMIS-ratings (scale 0 - 4)
HEALTH
FIRE
REACTIVITY
2
2
0
Health = 2
Fire = 2
Reactivity = 0
Other hazards
Results of PBT and vPvB assessment
PBT: N/A
vPvB: N/A
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Chemical characterization: Mixtures
Description: Mixture of the substances listed below with nonhazardous additions.
Dangerous components:
136-52-7 cobalt bis(2-ethylhexanoate)
SECTION 4. FIRST AID MEASURES
Description of first aid measures
General information:
Symptoms of poisoning may even occur after several hours; therefore medical observation for at least 48 hours
after the accident.
If inhaled: Supply fresh air; consult doctor in case of complaints.
In case of skin contact: Generally the product does not irritate the skin.
In case of eye contact: Rinse opened eye for several minutes under running water.
If swallowed: Immediately call a doctor.
Most important symptoms and effects, both acute and delayed:
No data available
Indication of any immediate medical attention and special treatment needed
No data available
SECTION 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing agents: CO2, sand, extinguishing powder. Do not use water.
For safety reasons unsuitable extinguishing agents: Water with full jet
Special hazards arising from the substance or mixture No data available
Advice for firefighters
Protective equipment: No special measures required.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Keep unprotected persons away.
Environmental precautions: No special measures required.
Methods and materials for containment and cleanup:
Absorb with liquid-binding material (sand, diatomite, acid binders, universal binders, sawdust).
Dispose contaminated material as waste according to item 13.
Ensure adequate ventilation.
Do not flush with water or aqueous cleansing agents
Reference to other sections
See Section 7 for information on safe handling.
See Section 8 for information on personal protection equipment.
See Section 13 for disposal information.
SECTION 7. HANDLING AND STORAGE
Precautions for safe handling
Ensure good ventilation/exhaustion at the workplace.
Prevent formation of aerosols.
Information about protection against explosions and fires:
Keep ignition sources away - Do not smoke.
Protect against electrostatic charges.
Conditions for safe storage, including any incompatibilities:
Requirements to be met by storerooms and receptacles: No special requirements.
Information about storage in one common storage facility: Not required.
Further information about storage conditions: Keep receptacle tightly sealed.
Specific end use(s) No data available
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Additional information about design of technical systems: No further data; see item 7.
Control parameters
Components with limit values that require monitoring at the workplace:
136-52-7 cobalt bis(2-ethylhexanoate)
TLV Long-term value: 0.02 mg/m3
as Co, BEI
Additional information: The lists that were valid during the creation were used as basis.
Exposure controls
Personal protective equipment:
General protective and hygienic measures:
Keep away from foodstuffs, beverages and feed.
Wash hands before breaks and at the end of work.
Breathing equipment:
In case of brief exposure or low pollution use respiratory filter device. In case of intensive or longer exposure use
respiratory protective device that is independent of circulating air.
Protection of hands:
The glove material has to be impermeable and resistant to the product/ the substance/ the preparation.
Due to missing tests no recommendation to the glove material can be given for the product/ the preparation/ the
chemical mixture.
Selection of the glove material on consideration of the penetration times, rates of diffusion and the degradation
Material of gloves
The selection of the suitable gloves does not only depend on the material, but also on further marks of quality and
varies from manufacturer to manufacturer. As the product is a preparation of several substances, the resistance
of the glove material can not be calculated in advance and has therefore to be checked prior to the application.
Penetration time of glove material
The exact break through time has to be found out by the manufacturer of the protective gloves and has to be
observed.
Eye protection:
Tightly sealed goggles
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
Appearance:
Form: Liquid
Color: Violet
Odor: Characteristic
Odor threshold: No data available.
pH: No data available.
Melting point/Melting range: Undetermined.
Boiling point/Boiling range: Undetermined.
Flash point: 40 °C (104 °F)
Flammability (solid, gas): N/A
Ignition temperature: 229 °C (444 °F)
Decomposition temperature: No data available.
Autoignition: Product is not selfigniting.
Danger of explosion: Product is not explosive. However, formation of explosive air/vapor
mixtures are possible.
Explosion limits:
Lower: 0.7 Vol %
Upper: 8.9 Vol %
Vapor pressure: No data available.
Density: No data available.
Relative density No data available.
Vapor density No data available.
Evaporation rate No data available.
Solubility in / Miscibility with
Water: Not miscible or difficult to mix.
Partition coefficient (n-octanol/water): No data available.
Viscosity:
Dynamic: No data available.
Kinematic: No data available.
Solvent content:
Organic solvents: 0.0 %
Other information No data available
SECTION 10. STABILITY AND REACTIVITY
Reactivity
Chemical stability
Thermal decomposition / conditions to be avoided: No decomposition if used according to specifications.
Possibility of hazardous reactions No dangerous reactions known.
Conditions to avoid No data available
Incompatible materials: No data available
Hazardous decomposition products: No dangerous decomposition products known.
SECTION 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity:
Primary irritant effect:
on the skin: No irritant effect.
on the eye: No irritating effect.
Sensitization: No sensitizing effects known.
Additional toxicological information:
The product shows the following dangers according to internally approved calculation methods for preparations:
Harmful
Carcinogenic categories
IARC (International Agency for Research on Cancer)
136-52-7 cobalt bis(2-ethylhexanoate) 2B
NTP (National Toxicology Program)
None of the ingredients is listed.
OSHA-Ca (Occupational Safety & Health Administration)
None of the ingredients is listed.
SECTION 12. ECOLOGICAL INFORMATION
Toxicity
Aquatic toxicity: No data available
Persistence and degradability No data available
Bioaccumulative potential No data available
Mobility in soil No data available
Additional ecological information: Not known to be hazardous to water.
Results of PBT and vPvB assessment
PBT: N/A
vPvB: N/A
Other adverse effects No data available
SECTION 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Recommendation:
Must not be disposed of together with household garbage. Do not allow product to enter drains, sewage systems, or other water courses system.
Uncleaned packagings:
Recommendation: Disposal must be made according to official regulations.
SECTION 14. TRANSPORT INFORMATION
UN-Number
DOT, IMDG, IATA UN1993
UN proper shipping name
DOT Flammable liquids, n.o.s.
IMDG, IATA FLAMMABLE LIQUID, N.O.S.
Transport hazard class(es)
DOT
Class 3 Flammable liquids
Label 3
IMDG, IATA
Class 3 Flammable liquids
Label 3
Packing group
DOT, IMDG, IATA III
Environmental hazards:
Marine pollutant: No
Special precautions for user Warning: Flammable liquids
EMS Number: F-E,S-E
Transport in bulk according to Annex II of
MARPOL73/78 and the IBC Code N/A
Transport/Additional information:
DOT
Quantity limitations On passenger aircraft/rail: 60 L
On cargo aircraft only: 220 L
IMDG
Limited quantities (LQ) 5L
Excepted quantities (EQ) Code: E1
Maximum net quantity per inner packaging: 30 ml
Maximum net quantity per outer packaging: 1000 ml
UN "Model Regulation": UN1993, Flammable liquids, n.o.s., 3, III
SECTION 15. REGULATORY INFORMATION
Safety, health and environmental regulations/legislation specific for the substance or mixture
Sara
Section 355 (extremely hazardous substances):
None of the ingredients is listed.
Section 313 (Specific toxic chemical listings):
All ingredients are listed.
TSCA (Toxic Substances Control Act):
All ingredients are listed.
Proposition 65
Chemicals known to cause cancer:
None of the ingredients is listed.
Chemicals known to cause reproductive toxicity for females:
None of the ingredients is listed.
Chemicals known to cause reproductive toxicity for males:
None of the ingredients is listed.
Chemicals known to cause developmental toxicity:
None of the ingredients is listed.
Carcinogenic categories
EPA (Environmental Protection Agency)
None of the ingredients is listed.
TLV (Threshold Limit Value established by ACGIH)
None of the ingredients is listed.
NIOSH-Ca (National Institute for Occupational Safety and Health)
None of the ingredients is listed.
GHS label elements The product is classified and labeled according to the Globally Harmonized System (GHS).
Hazard pictograms
~GHS02~GHS06~GHS08
Signal word Danger
Hazard-determining components of labeling:
cobalt bis(2-ethylhexanoate)
Hazard statements
H226 Flammable liquid and vapor.
H301 Toxic if swallowed.
H351 Suspected of causing cancer.
Precautionary statements
P101 If medical advice is needed, have product container or label at hand.
P102 Keep out of reach of children.
P103 Read label before use.
P210 Keep away from heat/sparks/open flames/hot surfaces. - No smoking.
P280 Wear protective gloves/protective clothing/eye protection/face protection.
P301+P310 IF SWALLOWED: Immediately call a POISON CENTER/ doctor.
P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if
present and easy to do. Continue rinsing.
P403+P233 Store in a well-ventilated place. Keep container tightly closed.
P501 Dispose of contents/container in accordance with local/regional/national/international
regulations.
Chemical safety assessment: A Chemical Safety Assessment has not been carried out.
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 compounds).
compounds). 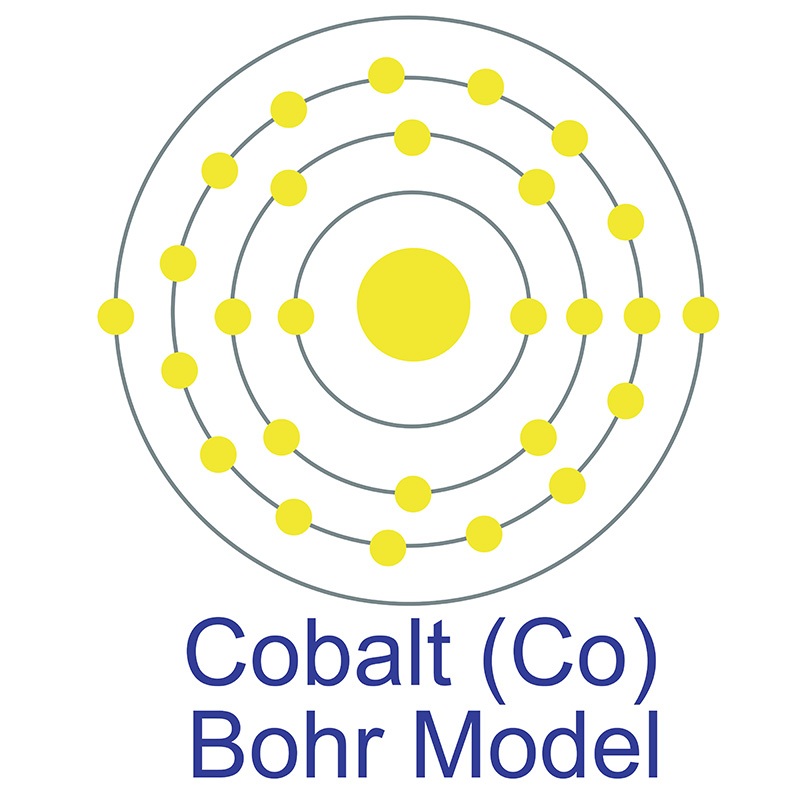 The number of electrons in each of cobalt's shells is 2, 8, 15, 2 and its electron configuration is [Ar]3d7 4s2. The cobalt atom has a radius of 125 pm and a Van der Waals radius of 192 pm. Cobalt was first discovered by George Brandt in 1732. In its elemental form, cobalt has a lustrous gray appearance. Cobalt is found in cobaltite, erythrite, glaucodot and skutterudite ores.
The number of electrons in each of cobalt's shells is 2, 8, 15, 2 and its electron configuration is [Ar]3d7 4s2. The cobalt atom has a radius of 125 pm and a Van der Waals radius of 192 pm. Cobalt was first discovered by George Brandt in 1732. In its elemental form, cobalt has a lustrous gray appearance. Cobalt is found in cobaltite, erythrite, glaucodot and skutterudite ores.  Cobalt produces brilliant blue pigments which have been used since ancient times to color paint and glass. Cobalt is a ferromagnetic metal and is used primarily in the production of
Cobalt produces brilliant blue pigments which have been used since ancient times to color paint and glass. Cobalt is a ferromagnetic metal and is used primarily in the production of