SECTION 1. IDENTIFICATION
Product Name: Nickel Zinc Iron Oxide Nanoparticles / Nanopowder
Product Number: All applicable American Elements product codes, e.g. NIZN-FEO-02-NP
, NIZN-FEO-03-NP
, NIZN-FEO-04-NP
, NIZN-FEO-05-NP
CAS #: 12645-50-0
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Classification of the substance or mixture
GHS Classification in accordance with 29 CFR 1910 (OSHA HCS)
Skin irritation (Category 2), H315
Eye irritation (Category 2A), H319
Skin sensitisation (Category 1), H317
Specific target organ toxicity - single exposure (Category 3), Respiratory system, H335
For the full text of the H-Statements mentioned in this Section, see Section 16.
GHS Label elements, including precautionary statements
Pictogram

Signal word: Warning
Hazard statement(s)
H315
Causes skin irritation.
H317
May cause an allergic skin reaction.
H319
Causes serious eye irritation.
H335
May cause respiratory irritation.
Precautionary statement(s)
P261
Avoid breathing dust/ fume/ gas/ mist/ vapours/ spray.
P264
Wash skin thoroughly after handling.
P271
Use only outdoors or in a well-ventilated area.
P272
Contaminated work clothing should not be allowed out of the workplace.
P280
Wear protective gloves/ eye protection/ face protection.
P302 + P352
IF ON SKIN: Wash with plenty of soap and water.
P304 + P340
IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
P305 + P351 + P338
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P312
Call a POISON CENTER/doctor if you feel unwell.
P321
Specific treatment (see supplemental first aid instructions on this label).
P333 + P313
If skin irritation or rash occurs: Get medical advice/ attention.
P337 + P313
If eye irritation persists: Get medical advice/ attention.
P362
Take off contaminated clothing and wash before reuse.
P403 + P233
Store in a well-ventilated place. Keep container tightly closed.
P405
Store locked up.
P501
Dispose of contents/ container to an approved waste disposal plant.
Hazards not otherwise classified (HNOC) or not covered by GHS - none
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substances
Synonyms:
Zinc nickel iron oxide
Nickel zinc ferrite
Zinc nickel ferrite
Formula: Fe4NiO4Zn
Molecular weight: 411.46 g/mol
CAS-No.: 12645-50-0
SECTION 4. FIRST AID MEASURES
Description of first aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
Move out of dangerous area.
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician.
In case of skin contact
Wash off with soap and plenty of water. Consult a physician.
In case of eye contact
Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician.
If swallowed
Never give anything by mouth to an unconscious person. Rinse mouth with water. Consult a physician.
Most important symptoms and effects, both acute and delayed
The most important known symptoms and effects are described in the labelling (see section 2.2) and/or in section 11
Indication of any immediate medical attention and special treatment needed
No data available
SECTION 5. FIREFIGHTING MEASURES
Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
Special hazards arising from the substance or mixture
No data available
Advice for firefighters
Wear self-contained breathing apparatus for firefighting if necessary.
Further information
No data available
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust.
For personal protection see section 8.
Environmental precautions
Do not let product enter drains.
Methods and materials for containment and cleaning up
Pick up and arrange disposal without creating dust. Sweep up and shovel. Keep in suitable, closed containers for disposal.
Reference to other sections
For disposal see section 13.
SECTION 7. HANDLING AND STORAGE
Exposure controls
Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and at the end of workday.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Exposure controls
Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and at the end of workday.
Personal protective equipment
Eye/face protection
Face shield and safety glasses Use equipment for eye protection tested and approved under appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protection
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique (without touching glove's outer surface) to avoid skin contact with this product. Dispose of contaminated gloves after use in accordance with applicable laws and good laboratory practices. Wash and dry hands.
Body Protection
Complete suit protecting against chemicals, The type of protective equipment must be selected according to the concentration and amount of the dangerous substance at the specific workplace.
Respiratory protection
For nuisance exposures use type P95 (US) or type P1 (EU EN 143) particle respirator.For higher level protection use type OV/AG/P99 (US) or type ABEK-P2 (EU EN 143) respirator cartridges. Use respirators and components tested and approved under appropriate government standards such as NIOSH (US) or CEN (EU).
Control of environmental exposure
Do not let product enter drains.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Information on basic physical and chemical properties
Appearance
Form: powder
Colour: dark brown, black
Odour
No data available
Odour Threshold
No data available
pH
No data available
Melting point/freezing point
No data available
Initial boiling point and boiling range
No data available
Flash point
Not applicable
Evaporation rate
No data available
Flammability (solid, gas)
No data available
Upper/lower flammability or explosive limits
No data available
Vapour pressure
No data available
Vapour density
No data available
Relative density
2.81 g/mL at 25 °C (77 °F)
Water solubility
insoluble
Partition coefficient: n-octanol/water
No data available
Auto-ignition temperature
No data available
Decomposition temperature
No data available
Viscosity
No data available
Explosive properties
No data available
Oxidizing properties
No data available
Other safety information
No data available
SECTION 10. STABILITY AND REACTIVITY
Reactivity
No data available
Chemical stability
Stable under recommended storage conditions.
Possibility of hazardous reactions
No data available
Conditions to avoid
No data available
Incompatible materials
Strong oxidizing agents
Hazardous decomposition products
Hazardous decomposition products formed under fire conditions.
- Nickel/nickel oxides, Iron oxides, Zinc/zinc oxides
Other decomposition products
- No data available
In the event of fire: see section 5
SECTION 11. TOXICOLOGICAL INFORMATION
Information on toxicological effects
Acute toxicity
No data available
Dermal:
No data available
No data available
Skin corrosion/irritation
No data available
Serious eye damage/eye irritation
No data available
Respiratory or skin sensitisation
Germ cell mutagenicity
No data available
Carcinogenicity
May cause cancer by inhalation.
IARC: 1-Group 1: Carcinogenic to humans (Nickel zinc ferrite)
IARC: 1-Group 1: Carcinogenic to humans (Nickel zinc ferrite)
NTP: Known-Known to be human carcinogen (Nickel zinc ferrite)
NTP: Known-Known to be human carcinogen (Nickel zinc ferrite)
OSHA:
No component of this product present at levels greater than or equal to 0.1% is on OSHA’s
list of regulated carcinogens.
No component of this product present at levels greater than or equal to 0.1% is on OSHA’s
list of regulated carcinogens.
Reproductive toxicity
No data available
Reproductive toxicity-Rat-Inhalation
Paternal Effects: Spermatogenesis (including genetic material, sperm morphology,motility, and count).
Reproductive toxicity-Rat-Inhalation
Effects on Fertility: Pre-implantation mortality (e.g., reduction in numbe corpora lutea). Effects on Embryo or Fetus: Fetal death.
No data available
Specific target organ toxicity -single exposure
Inhalation-May cause respiratory irritation.
Specific target organ toxicity -repeated exposure
No data available
Aspiration hazard
No data available
Additional Information
RTECS: NO7118000
SECTION 12. ECOLOGICAL INFORMATION
Toxicity
No data available
Persistence and degradability
No data available
Bioaccumulative potential
No data available
Mobility in soil
No data available
Results of PBT and vPvB assessment
PBT/vPvB assessment not available as chemical safety assessment not required/not conducted
Other adverse effects
No data available
SECTION 13. DISPOSAL CONSIDERATIONS
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company.
Contact a licensed professional waste disposal service to dispose of this material.
Contaminated packaging
Dispose of as unused product.
SECTION 14. TRANSPORT INFORMATION
DOT (US)
Not dangerous goods
IMDG
Not dangerous goods
IATA
Not dangerous goods
SECTION 15. REGULATORY INFORMATION
SARA 302 Components
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.
SARA 313 Components
The following components are subject to reporting levels established by SARA Title III, Section 313:
Nickel zinc ferrite
CAS-No.12645-50-0
Revision Date 2015-07-08
SARA 311/312
Hazards
Acute Health Hazard, Chronic Health Hazard
Massachusetts Right To Know
Components
No components are subject to the Massachusetts Right to Know Act.
Pennsylvania Right To Know
Components
Nickel zinc ferrite
CAS-No.12645-50-0
Revision Date
2015-07-08
New Jersey Right To Know
Components
Nickel zinc ferrite
CAS-No.12645-50-0
Revision Date
2015-07-08
California Prop. 65
Components
WARNING! This product contains a chemical known to the
State of California to cause cancer.
Nickel zinc ferrite
CAS-No.12645-50-0
Revision Date
2007-09-28
WARNING! This product contains a chemical known to the
State of California to cause cancer.
Nickel zinc ferrite
CAS-No.12645-50-0
Revision Date
2007-09-28
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 Nickel Zinc Iron Oxide (NiZnFe4O4) Nanoparticles are typically < 50 nm (XRD). Nano Nickel Zinc Iron Oxide Particles are also available in passivated and in
Nickel Zinc Iron Oxide (NiZnFe4O4) Nanoparticles are typically < 50 nm (XRD). Nano Nickel Zinc Iron Oxide Particles are also available in passivated and in 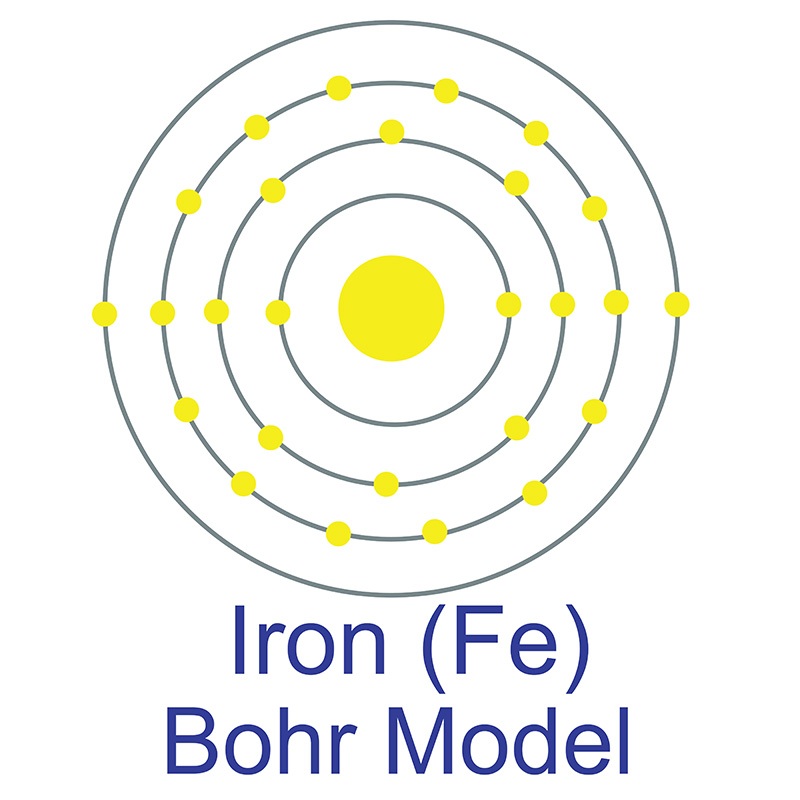 The iron atom has a radius of 126 pm and a Van der Waals radius of 194 pm. Iron was discovered by humans before 5000 BC. In its elemental form, iron has a lustrous grayish metallic appearance. Iron is the fourth most common element in the Earth's crust and the most common element by mass forming the earth as a whole. Iron is rarely found as a free element, since it tends to oxidize easily; it is usually found in minerals such as magnetite, hematite, goethite, limonite, or siderite.
The iron atom has a radius of 126 pm and a Van der Waals radius of 194 pm. Iron was discovered by humans before 5000 BC. In its elemental form, iron has a lustrous grayish metallic appearance. Iron is the fourth most common element in the Earth's crust and the most common element by mass forming the earth as a whole. Iron is rarely found as a free element, since it tends to oxidize easily; it is usually found in minerals such as magnetite, hematite, goethite, limonite, or siderite. Though pure iron is typically soft, the addition of carbon creates the
Though pure iron is typically soft, the addition of carbon creates the 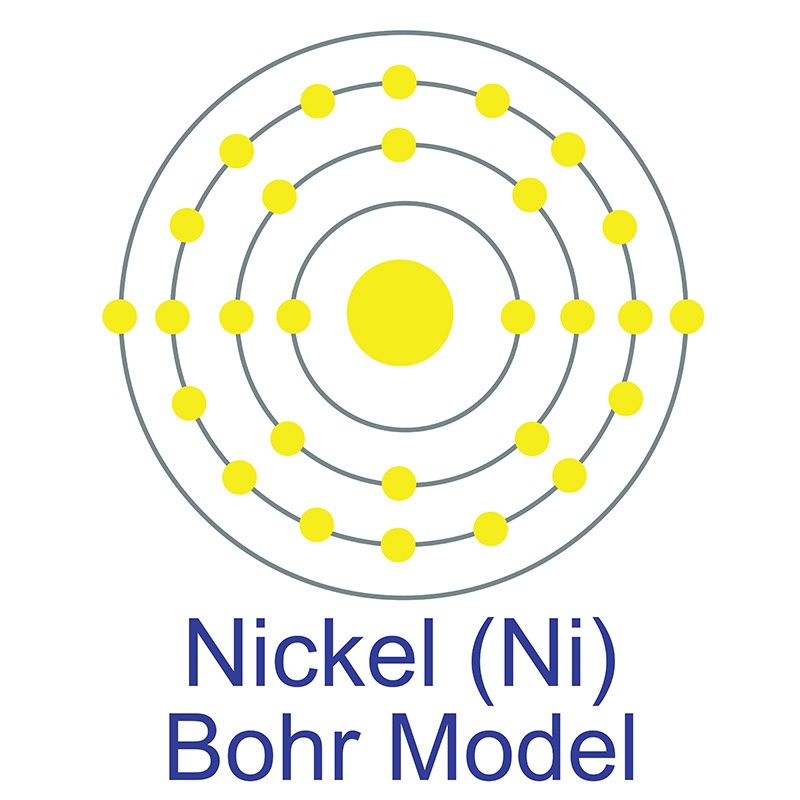 The number of electrons in each of nickel's shells is [2, 8, 16, 2] and its electron configuration is [Ar]3d8 4s2. Nickel was first discovered by Alex Constedt in 1751. The nickel atom has a radius of 124 pm and a Van der Waals radius of 184 pm. In its elemental form, nickel has a lustrous metallic silver appearance. Nickel is a hard and ductile
The number of electrons in each of nickel's shells is [2, 8, 16, 2] and its electron configuration is [Ar]3d8 4s2. Nickel was first discovered by Alex Constedt in 1751. The nickel atom has a radius of 124 pm and a Van der Waals radius of 184 pm. In its elemental form, nickel has a lustrous metallic silver appearance. Nickel is a hard and ductile  It is one of four elements that are ferromagnetic and is used in the production of various type of magnets for commercial use. Nickel is sometimes found free in nature but is more commonly found in ores. The bulk of mined nickel comes from laterite and magmatic sulfide ores. The name originates from the German word kupfernickel, which means "false
It is one of four elements that are ferromagnetic and is used in the production of various type of magnets for commercial use. Nickel is sometimes found free in nature but is more commonly found in ores. The bulk of mined nickel comes from laterite and magmatic sulfide ores. The name originates from the German word kupfernickel, which means "false 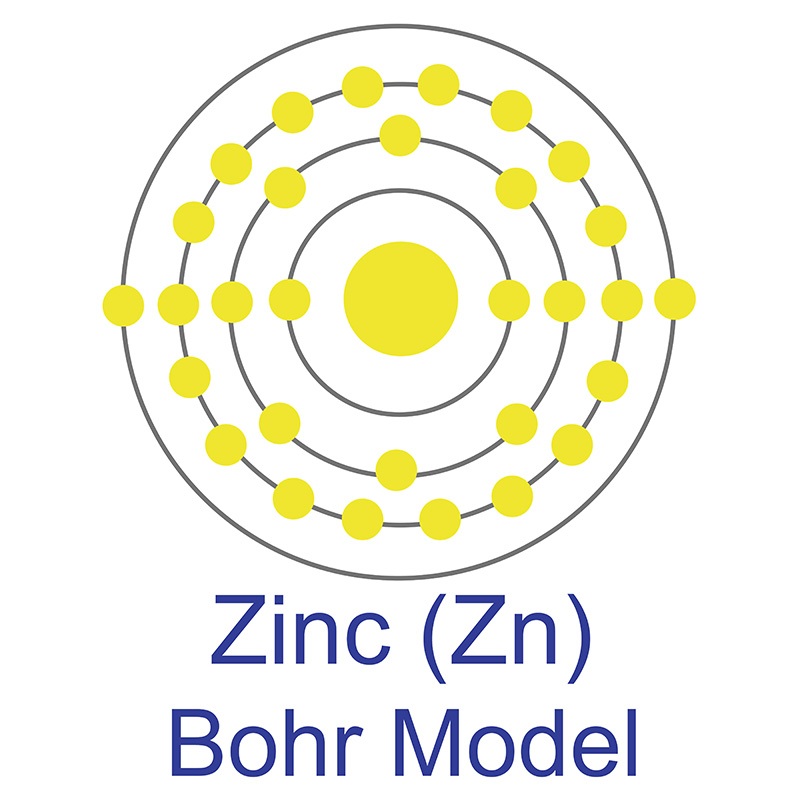 The zinc atom has a radius of 134 pm and a Van der Waals radius of 210 pm. Zinc was discovered by Indian
The zinc atom has a radius of 134 pm and a Van der Waals radius of 210 pm. Zinc was discovered by Indian  It is a fair conductor of electricity, and burns in air at high red producing white clouds of the
It is a fair conductor of electricity, and burns in air at high red producing white clouds of the 