SECTION 1. IDENTIFICATION
Product Name: Copper(II) Tetrafluoroborate Hexahydrate
Product Number: All applicable American Elements product codes, e.g. CU2-FBAT-018-C.6HYD
CAS #: 72259-10-0
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
This chemical is considered hazardous by the 2012 OSHA Hazard Communication Standard (29 CFR 1910.1200).
Classification
Skin corrosion/irritation Category 1 Sub-category A
Serious eye damage/eye irritation Category 1
Label elements
Signal word Danger
Hazard statements Causes severe skin burns and eye damage
Symbols/Pictograms

Precautionary Statements - Prevention Do not breathe dust/fume/gas/mist/vapors/spray
Wash face, hands and any exposed skin thoroughly after
handling
Wear protective gloves/protective clothing/eye protection/face
protection
Precautionary Statements - Response
Immediately call a POISON CENTER or doctor/physician
IF IN EYES: Rinse cautiously with water for several minutes.
Remove contact lenses, if present and easy to do. Continue
rinsing
Immediately call a POISON CENTER or doctor/physician
IF ON SKIN (or hair): Remove/Take off immediately all
contaminated clothing. Rinse skin with water/shower
Wash contaminated clothing before reuse
IF INHALED: Remove victim to fresh air and keep at rest in a
position comfortable for breathing
Immediately call a POISON CENTER or doctor/physician
IF SWALLOWED: Rinse mouth. DO NOT induce vomiting
Precautionary Statements - Storage Store locked up
Precautionary Statements - Disposal Dispose of contents/container to an approved waste disposal
plant
Hazards not otherwise classified (HNOC)
Hazards not otherwise classified (HNOC) N/A
Other Information
Unknown acute toxicity 100% of the mixture consists of ingredient(s) of unknown toxicity.
NFPA
Health hazards -
Flammability -
Stability -
Physical and chemical -
properties
HMIS
Health hazards 3
Flammability 0
Physical hazards 1
Personal protection -
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
CAS No 72259-10-0
Molecular Weight 345.25 (237.15anhy)
Formula Cu(BF4)2·6H2O
Chemical Name CAS No weight-% Oral LD50 dermal LD50 Inhalation LC50
Copper(II) tetrafluoroborate
hexahydrate
72259-10-0 >98 - - -
SECTION 4. FIRST AID MEASURES
First Aid Measures
General advice Consult a physician if necessary. Remove to fresh air.
Eye contact Wash with plenty of water.
Skin Contact Wash skin with soap and water.
Inhalation Remove to fresh air. If breathing is difficult, give oxygen. If not breathing, give artificial
respiration.
Ingestion Never give anything by mouth to an unconscious person. Clean mouth with water.
Most important symptoms and effects, both acute and delayed
Symptoms No data available.
Indication of any immediate medical attention and special treatment needed
Note to physicians Treat symptomatically.
SECTION 5. FIREFIGHTING MEASURES
Suitable Extinguishing Media
Suitable Extinguishing Media Use extinguishing measures that are appropriate to local circumstances and the
surrounding environment.
Unsuitable Extinguishing Media None.
Specific hazards arising from the chemical
Specific hazards arising from the
chemical
No data available.
Hazardous combustion products Hydrogen fluoride.
Explosion data
Sensitivity to Mechanical Impact No data available.
Sensitivity to Static Discharge No data available.
Protective equipment and precautions for firefighters
Protective equipment and precautions
for firefighters
As in any fire, wear self-contained breathing apparatus pressure-demand, MSHA/NIOSH
(approved or equivalent) and full protective gear.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and emergency procedures
Personal precautions Ensure adequate ventilation, especially in confined areas.
Environmental precautions
Environmental precautions See Section 12 for additional Ecological Information.
Methods and material for containment and cleaning up
Methods for containment Prevent further leakage or spillage if safe to do so.
Methods for cleaning up Use personal protective equipment as required. Cover powder spill with plastic sheet or tarp
to minimize spreading and keep powder dry. Take up mechanically, placing in appropriate
containers for disposal. Avoid creating dust. Clean contaminated surface thoroughly.
SECTION 7. HANDLING AND STORAGE
Precautions for safe handling
Advice on safe handling Handle in accordance with good industrial hygiene and safety practice.
Conditions for safe storage, including any incompatibilities Conditions Keep containers tightly closed in a dry, cool and well-ventilated place. Store at room
temperature.
Incompatible materials None known based on information supplied.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Control parameters
Exposure Guidelines This product, as supplied, does not contain any hazardous materials with occupational
exposure limits established by the region specific regulatory bodies.
Appropriate engineering controls
Engineering Controls Showers
Eyewash stations
Ventilation systems
Individual protection measures, such as personal protective equipment
Eye/face protection Wear safety glasses with side shields (or goggles).
Skin and Body Protection Wear protective gloves and protective clothing.
Respiratory protection If exposure limits are exceeded or irritation is experienced, NIOSH/MSHA approved
respiratory protection should be worn. Positive-pressure supplied air respirators may be
required for high airborne contaminant concentrations. Respiratory protection must be
provided in accordance with current local regulations.
General Hygiene Considerations Handle in accordance with good industrial hygiene and safety practice.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical State Solid
Appearance No data available
Odor No data available
Property Values
pH No data available
Melting point/freezing point No data available
Boiling point No data available
Flash point No data available
Density No data available
Evaporation rate No data available
Upper flammability limits No data available
Lower flammability limit No data available
Vapor pressure No data available
Vapor density No data available
Specific gravity No data available
Water solubility No data available
Solubility in other solvents No data available
Partition coefficient No data available
Autoignition temperature No data available
Decomposition temperature No data available
Kinematic viscosity No data available
Explosive properties No data available
Oxidizing properties No data available
SECTION 10. STABILITY AND REACTIVITY
Reactivity N/A
Chemical stability Stable under recommended storage conditions.
Possibility of Hazardous Reactions None under normal processing.
Hazardous polymerization No data available.
Conditions to avoid Extremes of temperature and direct sunlight.
Incompatible materials Strong oxidizing agents.
Hazardous Decomposition Products Hydrogen fluoride.
SECTION 11. TOXICOLOGICAL INFORMATION
Information on likely routes of exposure
Inhalation No data available.
Eye contact No data available.
Skin Contact No data available.
Ingestion No data available.
Information on toxicological effects
Symptoms No data available.
Delayed and immediate effects as well as chronic effects from short and long-term exposure
Chronic Toxicity No data available.
Numerical measures of toxicity - Product Information
Unknown acute toxicity 100% of the mixture consists of ingredient(s) of unknown toxicity
SECTION 12. ECOLOGICAL INFORMATION
Ecotoxicity May cause long lasting harmful effects to aquatic life
100% of the mixture consists of components(s) of unknown hazards to the aquatic environment.
Persistence and degradability No data available.
Bioaccumulation No data available.
Mobility No data available.
SECTION 13. DISPOSAL CONSIDERATIONS
Disposal of wastes Disposal should be in accordance with applicable regional, national and local laws and
regulations.
Contaminated packaging Do not reuse container.
US EPA Waste Number D002
SECTION 14. TRANSPORT INFORMATION
DOT
UN/ID no UN3260
Hazard Class 8
Packing Group III
Proper shipping name Corrosive solid, acidic, inorganic, n.o.s.*
Description UN3260, Corrosive solid, acidic, inorganic, n.o.s.*, 8, III
Emergency Response Guide Number 154
IMDG
UN/ID no UN3260
Hazard Class 8
Packing Group III
Proper shipping name Corrosive solid, acidic, inorganic, n.o.s.*
Description UN3260, Corrosive solid, acidic, inorganic, n.o.s.*, 8, III
Special Provisions IB8, IP3, T1, TP33
EmS-No F-A, S-B
IATA
UN/ID no UN3260
Hazard Class 8
Packing Group III
Proper shipping name Corrosive solid, acidic, inorganic, n.o.s.*
Description UN3260, Corrosive solid, acidic, inorganic, n.o.s.*, 8, III
SECTION 15. REGULATORY INFORMATION
International Inventories
All of the components in the product are on the following Inventory lists
No data available
X - Listed
TSCA - United States Toxic Substances Control Act Section 8(b) Inventory
DSL/NDSL - Canadian Domestic Substances List/Non-Domestic Substances List
EINECS/ELINCS - European Inventory of Existing Chemical Substances/European List of Notified Chemical Substances
ENCS - Japan Existing and New Chemical Substances
IECSC - China Inventory of Existing Chemical Substances
KECL - Korean Existing and Evaluated Chemical Substances
PICCS - Philippines Inventory of Chemicals and Chemical Substances
US Federal Regulations
SARA 313
Section 313 of Title III of the Superfund Amendments and Reauthorization Act of 1986 (SARA). This product does not contain any
chemicals which are subject to the reporting requirements of the Act and Title 40 of the Code of Federal Regulations, Part 372.
SARA 311/312 Hazard Categories
Acute health hazard No
Chronic Health Hazard No
Fire hazard No
Sudden release of pressure hazard No
Reactive hazard No
CWA (Clean Water Act)
This product does not contain any substances regulated as pollutants pursuant to the Clean Water Act (40 CFR 122.21 and 40
CFR 122.42).
US State Regulations
California Proposition 65
This product does not contain any Proposition 65 chemicals.
U.S. State Right-to-Know Regulations
This product does not contain any substances regulated by state right-to-know regulations
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 Boron (atomic symbol: B, atomic number: 5) is a Block P, Group 13, Period 2 element with an atomic weight of 10.81. The number of electrons in each of boron's shells is 2, 3 and its electron configuration is [He] 2s2 2p1. The boron atom has a radius of 90 pm and a Van der Waals radius of 192 pm. Boron was discovered by Joseph Louis Gay-Lussac and Louis Jacques Thénard in 1808 and was first isolated by Humphry Davy later that year. Boron is classified as a metalloid is not found naturally on earth.
Boron (atomic symbol: B, atomic number: 5) is a Block P, Group 13, Period 2 element with an atomic weight of 10.81. The number of electrons in each of boron's shells is 2, 3 and its electron configuration is [He] 2s2 2p1. The boron atom has a radius of 90 pm and a Van der Waals radius of 192 pm. Boron was discovered by Joseph Louis Gay-Lussac and Louis Jacques Thénard in 1808 and was first isolated by Humphry Davy later that year. Boron is classified as a metalloid is not found naturally on earth.  Along with carbon and nitrogen, boron is one of the few elements in the periodic table known to form stable compounds featuring triple bonds. Boron has an energy band gap of 1.50 to 1.56 eV, which is higher than that of either silicon or germanium. The name Boron originates from a combination of carbon and the Arabic word buraqu meaning borax.
Along with carbon and nitrogen, boron is one of the few elements in the periodic table known to form stable compounds featuring triple bonds. Boron has an energy band gap of 1.50 to 1.56 eV, which is higher than that of either silicon or germanium. The name Boron originates from a combination of carbon and the Arabic word buraqu meaning borax.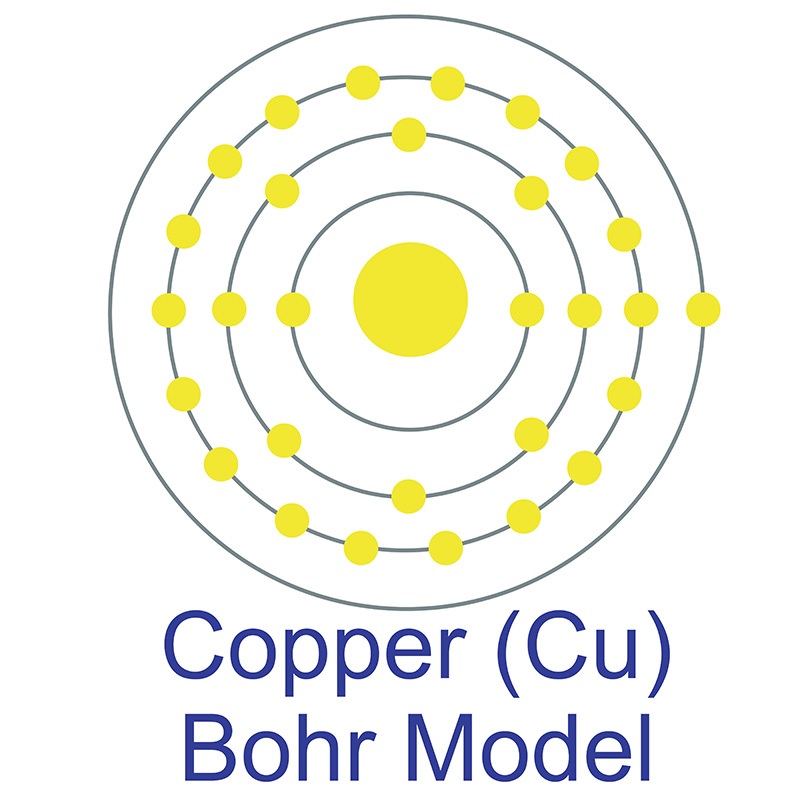 Copper (atomic symbol: Cu, atomic number: 29) is a Block D, Group 11, Period 4 element with an atomic weight of 63.546. The number of electrons in each of copper's shells is 2, 8, 18, 1 and its electron configuration is [Ar]3d10 4s1. The copper atom has a radius of 128 pm and a Van der Waals radius of 186 pm. Copper was first discovered by Early Man prior to 9000 BC. In its elemental form, copper has a reddish-orange metallic and lustrous appearance. Of all pure
Copper (atomic symbol: Cu, atomic number: 29) is a Block D, Group 11, Period 4 element with an atomic weight of 63.546. The number of electrons in each of copper's shells is 2, 8, 18, 1 and its electron configuration is [Ar]3d10 4s1. The copper atom has a radius of 128 pm and a Van der Waals radius of 186 pm. Copper was first discovered by Early Man prior to 9000 BC. In its elemental form, copper has a reddish-orange metallic and lustrous appearance. Of all pure  has a higher electrical conductivity. The origin of the word copper comes from the Latin word 'cuprium' which translates as "metal of Cyprus," as the Mediterranean island of Cyprus was known as an ancient source of mined copper..
has a higher electrical conductivity. The origin of the word copper comes from the Latin word 'cuprium' which translates as "metal of Cyprus," as the Mediterranean island of Cyprus was known as an ancient source of mined copper..