SECTION 1. IDENTIFICATION
Product Name: Copper Gallium Selenide Sputtering Target
Product Number: All applicable American Elements product codes, e.g. CU-GASE-04-ST
, CU-GASE-045-ST
, CU-GASE-05-ST
, CU-GASE-06-ST
CAS #: 12018-83-6
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Signal Word: Danger
Hazard Statements: H300+H330: Fatal if swallowed or if inhaled
H373: May cause damage to organs through prolonged or repeated exposure.
Precautionary Statements: P260: Do not breathe dust/fume/gas/mist/vapours/spray
P301+P310: IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician.
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P284: Wear respiratory protection
P320: Specific treatment is urgent (see on this label)
P405: Store locked up
P501: Dispose of contents/container in accordance with local/regional/national/international regulations
HMIS Health Ratings (0-4):
Health: 2
Flammability: 0
Physical: 1
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Chemical Family: Semimetallic alloy
Additional Names: Copper gallium dselenide
Copper gallium selenide (CuGaSe2):
Percentage: 100 wt%
CAS #: 12018-83-6
EC #: N/A
SECTION 4. FIRST AID MEASURES
General Treatment: Seek medical attention if symptoms persist.
Special Treatment: None
Important Symptoms: None
Inhalation: Remove victim to fresh air. Supply oxygen if breathing is difficult.
Ingestion: Seek medical attention
Skin: Wash affected area with mild soap and water. Remove any contaminated clothing.
Eyes: Flush eyes with water, blinking often for several minutes. Remove contact lenses if present and easy to do. Continue rinsing.
SECTION 5. FIREFIGHTING MEASURES
Flammability: Non-flammable
Extinguishing Media: Carbon dioxide, extinguishing powder or water spray. Fight larger fires with water spray or alcohol resistant foam.
Spec. Fire Fighting Procedure: Use full-face, self-contained breathing apparatus with full protective clothing to prevent contact with skin and eyes. See section 10 for decomposition products.
SECTION 6. ACCIDENTAL RELEASE MEASURES
If Material Is Released/Spilled: Wear appropriate respiratory and protective equipment specified in special protection information. Isolate spill area and provide ventilation. Vacuum up spill using a high efficiency particulate absolute (HEPA) air filter and place in a closed container for disposal. Take care not to raise dust.
Environmental Precautions: Isolate runoff to prevent environmental pollution.
SECTION 7. HANDLING AND STORAGE
Handling Conditions: Wash thoroughly after handling.
Storage Conditions: Store in a cool dry place in a tightly sealed container. Store apart from materials and conditions listed in section 10.
Work/Hygienic Maintenance: Do not use tobacco or food in work area. Wash thoroughly before eating and smoking. Do not blow dust off clothing or skin with compressed air.
Ventilation: Provide sufficient ventilation to maintain concentration at or below threshold limit.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
Permissible Exposure Limits: 0.1 mg/m3 as Cu, long-term value
Threshold Limit Value: 0.2 mg/m3 as Se, long-term value
Special Equipment: None
Respiratory Protection: Dust Respirator
Protective Gloves: Rubber gloves
Eye Protection: Safety glasses or goggles
Body Protection: Protective work clothing. Wear close-toed shoes and long sleeves/pants.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
Color Steel grey
Form: Powder, Granules, Pellets, Sputtering target, Custom parts
Odor: Odorless
Water Solubility: Insoluble
Boiling Point: N/A
Melting Point: N/A
Flash Point: N/A
Autoignition Temperature: N/A
Density: N/A
Molecular weight: 291.21 g/mol
SECTION 10. STABILITY AND REACTIVITY
Stability: Stable under recommended storage conditions
Reacts With: Oxidizing agents
Incompatible Conditions: None
Hazardous Decomposition Products: Metal oxide fume, Selenium dioxide, Hydrogen selenide
SECTION 11. TOXICOLOGICAL INFORMATION
Potential Health Effects:
Eyes: Causes irritating effect
Skin: Irritant to skin and mucous membranes
Ingestion: May cause irritation
Inhalation: May cause irritation
Chronic: Selenium may cause amyotropic lateral sclerosis, bronchial irritation, gastrointestinal distress, vasopharyngeal irritation, garlic odor on breath and sweat, metallic taste, pallor, irritability, excessive fatigue, loss of fingernails and hair, pulmonary edema, anemia and weight loss.
Copper compounds may be irritating to the skin, eyes and respiratory tract. They may cause metal fume fever, hemolysis of the red blood cells and injury to the liver, lungs, kidneys, and pancreas. Ingestion may also cause vomiting, gastric pain, dizziness, anemia, cramps, convulsions, shock, coma, and death.
Signs & Symptoms: N/A
Aggravated Medical Conditions: N/A
Median Lethal Dose: N/A
Carcinogen: IARC-3: Not classifiable as to carcinogenicity to humans.
SECTION 12. ECOLOGICAL INFORMATION
Aquatic Toxicity: High
Persistent Bioaccumulation Toxicity: No
Very Persistent, Very Bioaccumulative: No
Notes: Very toxic for aquatic organism.
May cause long lasting harmful effect on aquatic life.
Do not allow material to be released to the environment without proper governmental permits.
Do not allow product to reach any water sources.
Danger to drinking water if even extremely small quantities leak into the ground.
Also poisonous for fish and plankton in water bodies.
Avoid transfer into the environment.
SECTION 13. DISPOSAL CONSIDERATIONS
Dispose of in accordance with local, state, national, and international regulations.
SECTION 14. TRANSPORT INFORMATION
Hazardous: Hazardous for transportation.
Hazard Class: 9 Miscellaneous hazardous substances
Packing Group: III
UN Number: UN3077
Proper Shipping Name: Environmentally hazardous substances, solid, n.o.s. (Copper gallium selenide)
SECTION 15. REGULATORY INFORMATION
Sec 302 Extremely Hazardous: No
Sec 304 Reportable Quantities: N/A
Sec 313 Toxic Chemicals: Yes
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.

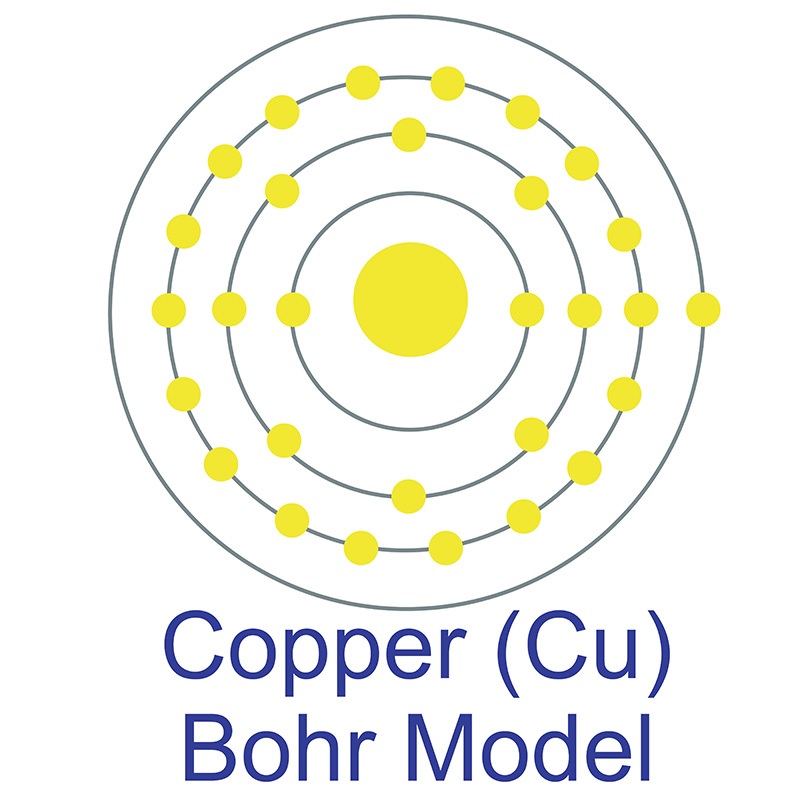 Copper (atomic symbol: Cu, atomic number: 29) is a Block D, Group 11, Period 4 element with an atomic weight of 63.546. The number of electrons in each of copper's shells is 2, 8, 18, 1 and its electron configuration is [Ar]3d10 4s1. The copper atom has a radius of 128 pm and a Van der Waals radius of 186 pm. Copper was first discovered by Early Man prior to 9000 BC. In its elemental form, copper has a reddish-orange metallic and lustrous appearance. Of all pure
Copper (atomic symbol: Cu, atomic number: 29) is a Block D, Group 11, Period 4 element with an atomic weight of 63.546. The number of electrons in each of copper's shells is 2, 8, 18, 1 and its electron configuration is [Ar]3d10 4s1. The copper atom has a radius of 128 pm and a Van der Waals radius of 186 pm. Copper was first discovered by Early Man prior to 9000 BC. In its elemental form, copper has a reddish-orange metallic and lustrous appearance. Of all pure  has a higher electrical conductivity. The origin of the word copper comes from the Latin word 'cuprium' which translates as "metal of Cyprus," as the Mediterranean island of Cyprus was known as an ancient source of mined copper..
has a higher electrical conductivity. The origin of the word copper comes from the Latin word 'cuprium' which translates as "metal of Cyprus," as the Mediterranean island of Cyprus was known as an ancient source of mined copper..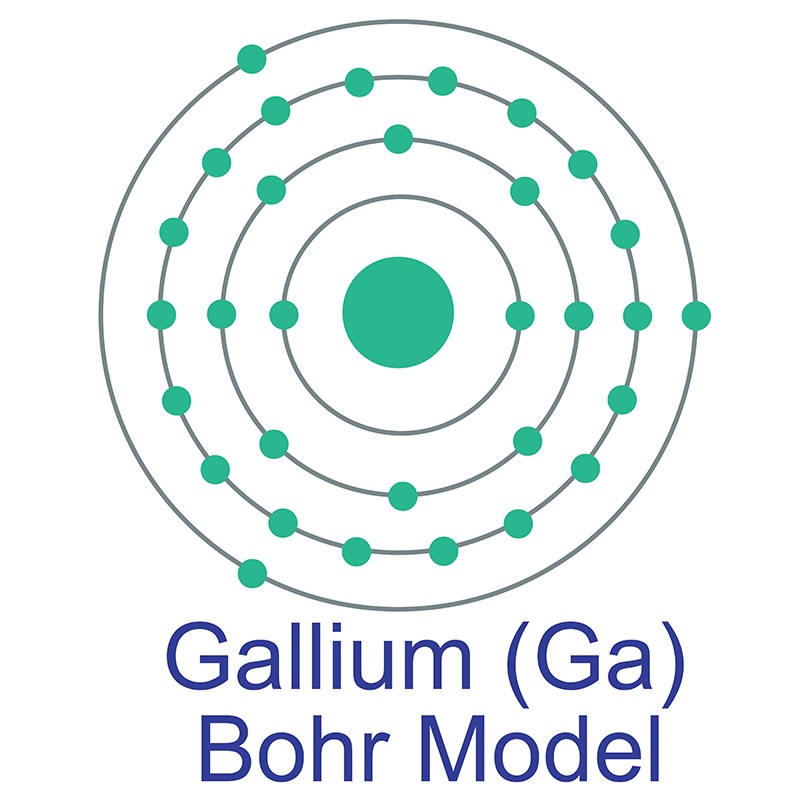 Gallium was predicted by Dmitri Mendeleev in 1871. It was first discovered and isolated by Lecoq de Boisbaudran in 1875. In its elemental form, gallium has a silvery appearance.
Gallium was predicted by Dmitri Mendeleev in 1871. It was first discovered and isolated by Lecoq de Boisbaudran in 1875. In its elemental form, gallium has a silvery appearance.  Gallium is one of three elements that occur naturally as a liquid at room temperature, the other two being
Gallium is one of three elements that occur naturally as a liquid at room temperature, the other two being 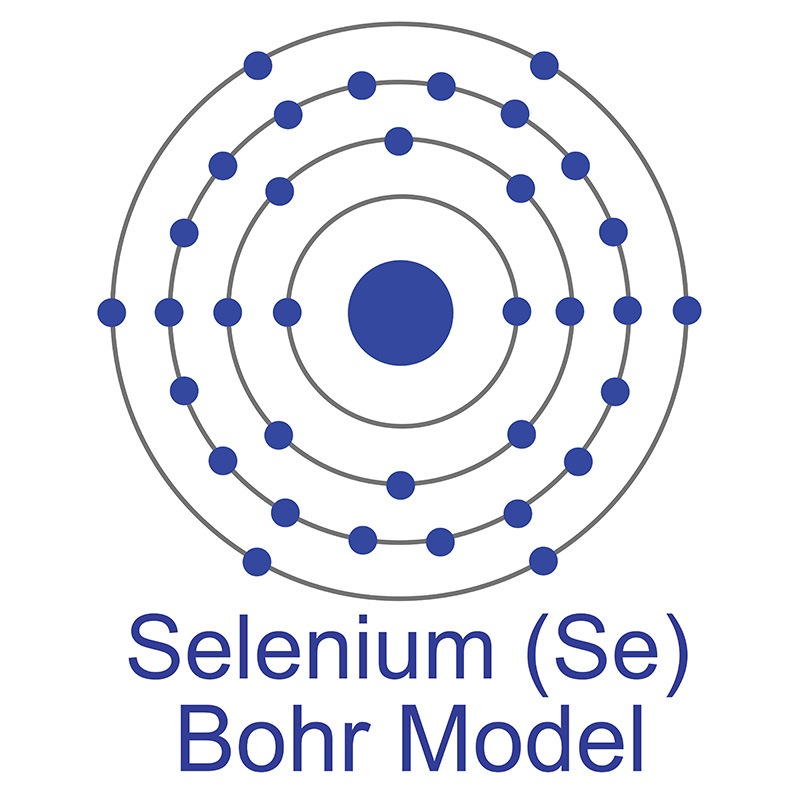 See more Selenium products.
See more Selenium products. One of the most common uses for selenium is in
One of the most common uses for selenium is in 
