SECTION 1. IDENTIFICATION
Product Name: Sodium Gold(I) Sulfite Solution
Product Number: All applicable American Elements product codes, e.g. NAAU-SIT-01-SOL
CAS #: 19153-98-1
Relevant identified uses of the substance: Scientific research and development
Supplier details:
American Elements
10884 Weyburn Ave.
Los Angeles, CA 90024
Tel: +1 310-208-0551
Fax: +1 310-208-0351
Emergency telephone number:
Domestic, North America: +1 800-424-9300
International: +1 703-527-3887
SECTION 2. HAZARDS IDENTIFICATION
Signal Word: Danger
Hazard Category:
Corrosive to metals Cat 1
Skin corrosion/irritation Cat 1
Acute tox, oral Cat 4
Acute tox, inh. Cat 4


Hazard Statements:
H290: May be corrosive to metals
H314: Causes severe skin burns and eye damage
H302: Harmful if swallowed
H332: Harmful if inhaled
P260: Do not breathe dust/fume/gas/mist/vapors/spray
P303+361+353: IF ON SKIN (or hair): Remove/Take off immediately all contaminated
P305+351+338: IF IN EYES: Rinse cautiously with water for several minutes. Remove
P301+330+331: IF SWALLOWED: Rinse mouth. Do NOT induce vomiting
P405: Store locked up
P501: Dispose of contents/container in accordance with local/regional/national/international rules
Hazards not otherwise classified:
To the best of our knowledge the chemical, physical, and toxicological properties of this
compound have not been thoroughly investigated. Avoid elevated temperature.
Incompatible with most metals, alkalines, metallic oxides, strong oxidants. Also water
reactive materials i.e. conc. sulfuric acid, oleum & acetic
SECTION 3. COMPOSITION/INFORMATION ON INGREDIENTS
Hazardous substance / CAS / Weight%:
Sodium gold (I) sulfite solution / 19153-98-1 / 99-100
SECTION 4. FIRST AID MEASURES
General Info: Ensure proper ventilation.
Notable Exposure symptoms: Irritation to skin, eyes and areas of contact
If ingested: Rinse mouth. Seek immediate medical attention. Clear the airway and administer artificial respiration if not breathing.
If swallowed, do not induce vomiting unless directed to do so by medical personnel.
If inhaled: Remove the victim from the contaminated area while protecting yourself from exposure by wearing an appropriate respirator. Put a similar respirator on the victim if possible. Get medical attention immediately.
Eye contact: Immediately flush eyes with plenty of water for at least 15 minutes. Remove contact lenses if present and easy to do. Get medical attention immediately.
Skin contact: Take off immediately all contaminated clothing. Rinse with water thoroughly. Get medical attention immediately.
Additional info: Observe for any symptoms for several hours after exposure. Follow up with medical attention if symptoms develop.
SECTION 5. FIREFIGHTING MEASURES
General info: Use proper safety equipment. If this product is involved in a fire Sodium sulfite and metal oxide fume can be released.
Extinguishing Method / Equipment: Use water spray or dry chemical, carbon dioxide. DO NOT use organics
Hazardous Decomposition Info: Constituents associated with burning / combustion are to be considered toxic. Firefighters should
wear self-contained breathing apparatus and full protective gear.
Use NIOSH/MSHA approved self-contained breathing apparatus and full protective clothing.
EMITS TOXIC FUMES UNDER FIRE CONDITIONS.
SECTION 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment and procedures: Keep unnecessary personnel away. Keep people away from and upwind of spill/leak. Eliminate all
ignition sources (no smoking, flares, sparks, or flames in immediate area). Wear appropriate
protective equipment (SCBA) and clothing during clean-up. Avoid breathing vapor. Ventilate
area if easy to do so and wash spill site after clean up is complete. Contact local authorities if
significant spillages cannot be contained. For personal protection, see section 8 of the SDS.
Containment Equipment and Procedures: Large Spills: Dike major spills. Absorb with liquid-binding material (sand, diatomite, acid binders,
universal binders, sawdust). Use neutralizing agent. Dispose contaminated material as waste. Ensure adequate ventilation
Cleanup procedures: Collect all contaminated media, or other cleanup materials into a waste receptacle. If cleaning surface is necessary, utilize vacuum cleaner, provided adequate ventilation is available.
SECTION 7. HANDLING AND STORAGE
Safe Handling Precautions: Do not handle until all safety precautions have been read and understood. Keep containers tightly closed Ensure adequate ventilation. Avoid breathing vapor. Avoid contact with eyes, skin, and clothing. Avoid prolonged exposure. Wear appropriate personal protective equipment and wash thoroughly after handling.
Work/Hygienic Practices: Always use good personal hygiene after handling
Recommendations for Storage: Store in the dark. Store away from strong bases. Keep from freezing. Keep container upright
Other precautions: 0.01 ppm high efficiency particular respirator/supplied air respirator/self-contained breathing apparatus; 0.2 ppm same as above with full face piece; 1 ppm powdered air purifying respirator with high efficiency filter/type C supplied air respirator operated in pressure demand mode. Store in a sealed container, plastic or plastic liner, in a cool, well ventilated, dry area away from volatile acids.
SECTION 8. EXPOSURE CONTROLS/PERSONAL PROTECTION
General / Engineering Controls: Ventilation: Local Exhaust: Required in handling area
Mechanical: Desirable to insure concentration of material below TLV/TWA levels
Other: Closed ventilation system (Laboratory Fume Hood)
Work Clothing: Protective work clothing which covers skin and prevents exposures. Lab coat/apron, flame and chemical resistant protective clothing, eye wash, safety shower, and hygiene facilities for washing.
Eye/face protection: Wear safety glasses with side shields or goggles or face shield
Skin Protection: Wear chemical resistant gloves.
Respiratory Protection: NIOSH approved chemical cartridge respirator for acid gas/chlorine (42 CFR part 84) and selfcontained breathing apparatus with full face shield.
SECTION 9. PHYSICAL AND CHEMICAL PROPERTIES
State: Liquid
Melting Point: N/A
Freezing Point: N/A
Color: Yellow Solution
Boiling Point/Range: 105 °C
pH 8 - 10
Molecular Wt. 424.06
Odor: Slight sulfur odor
Water Solubility: soluble
Evaporation rate: N/A
Flash Point: N/A
Part. Coeff (noctanol/water
Upper Flam Limits: N/A
Lower Flam Limits: N/A
Vapor Pressure: N/A
VOC Content (lbs/gal): N/A
Viscosity: N/A
Autoignition Temp: N/A
Specific Gravity: N/A
SECTION 10. STABILITY AND REACTIVITY
General: This product is stable and non-reactive under normal conditions of use. Product is not subject to hazardous polymerization. Avoid open flame and ignition sources. Conditions To Avoid (Stability) Elevated temperature.
Incompatible materials: Most metals, alkalines, metallic oxides, strong oxidants. Also water reactive materials i.e conc. sulfuric acid, oleum & acetic anhydrate. Carbonates, cyanides and sulfides
Decomposition products: Sulfur Dioxide Decomposition will not occur if used and stored according to specifications
SECTION 11. TOXICOLOGICAL INFORMATION
General Information: Harmful if swallowed. Swallowing will lead to a strong corrosive effect on mouth and throat and to the danger of perforation of esophagus and stomach. The RTECS contains acute toxicity data for this product
Toxicological Information (product): To the best of our knowledge, the chemical, physical, and toxicological properties of this compound have not been thoroughly investigated.
Likely Routes of Exposure: Eye Contact Skin Contact Inhalation Ingestion - possible, but considered unlikely.
Carcinogenicity or mutagenicity: No data available
Sensitization: Product is not expected to be a sensitizer.
SECTION 12. ECOLOGICAL INFORMATION
General information: This product is manufactured in accordance to and in compliance with all aspects of the Toxic Substances Control Act (TSCA) and that all compounds are on the TSCA inventory; This product is also in compliance with the New Substances Notification Regulations (NSNR) and all ingredients are on the DSL/NDSL.
Aquatic toxicity: No Data Available
Degradation / Mobility info: No Data Available
Bioaccumulative potential: No Data Available
SECTION 13. DISPOSAL CONSIDERATIONS
Product disposal: Consult Federal EPA, State and local regulations for proper disposal/recycle/reclamation
Container disposal: Treat empty containers with extra care. Consult waste contractor.
Other considerations: NOTE: Chemical additions, processing, or otherwise altering this material may make the waste management information presented above incomplete, inaccurate, or otherwise inappropriate.
SECTION 14. TRANSPORT INFORMATION
DOT: roper Shipping Name
Corrosive liquid n.o.s.
Hazard Class
8
DOT Identification Number
1760
DOT Shipping Label
Corrosive liquid n.o.s.
Packaging Requirements
See DOT Regulations
IMO/IMDG: Inquire for further transport information
IATA: Same as DOT, always consult IATA manual before shipping
SECTION 15. REGULATORY INFORMATION
TSCA: All ingredients of this product are listed under the U.S. Toxic Substances Control Act (TSCA)
Chemical Substance Inventory.
Canadian Regulatory Information: This product has been classified in accordance with the hazard criteria of the CPR, and the MSDS contains all the information required by the CPR.
European Union (EU) Regulatory Information: Symbol of Danger: C
Indication of Danger: Corrosive
R: 34, 36, 37
Risk Statements: Causes burns, Irritating to eyes, Irritating to respiratory system.
S: 17 26 27 36/37 39 45
Safety Statements: Keep away from combustible material. In case of contact with eyes, rinse
immediately with plenty of water and seek medical advice. Take off immediately all contaminated
clothing. Wear suitable protective clothing, gloves, and eye/face protection. In case of accident or if
you feel unwell, seek medical advice immediately.
SECTION 16. OTHER INFORMATION
Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH). The above information is believed to be correct but does not purport to be all inclusive and shall be used only as a guide. The information in this document is based on the present state of our knowledge and is applicable to the product with regard to appropriate safety precautions. It does not represent any guarantee of the properties of the product. American Elements shall not be held liable for any damage resulting from handling or from contact with the above product. See reverse side of invoice or packing slip for additional terms and conditions of sale. COPYRIGHT 1997-2022 AMERICAN ELEMENTS. LICENSED GRANTED TO MAKE UNLIMITED PAPER COPIES FOR INTERNAL USE ONLY.
 The gold atom has a radius of 144 pm and a Van der Waals radius of 217 pm. Gold was first discovered by Early Man prior to 6000 B.C. In its elemental form, gold has a metallic yellow appearance. Gold is a soft
The gold atom has a radius of 144 pm and a Van der Waals radius of 217 pm. Gold was first discovered by Early Man prior to 6000 B.C. In its elemental form, gold has a metallic yellow appearance. Gold is a soft  It is a good conductor of heat and electricity, and is unaffected by air and most reagents. It is one of the least reactive chemical elements. Gold is often found as a free element and with
It is a good conductor of heat and electricity, and is unaffected by air and most reagents. It is one of the least reactive chemical elements. Gold is often found as a free element and with 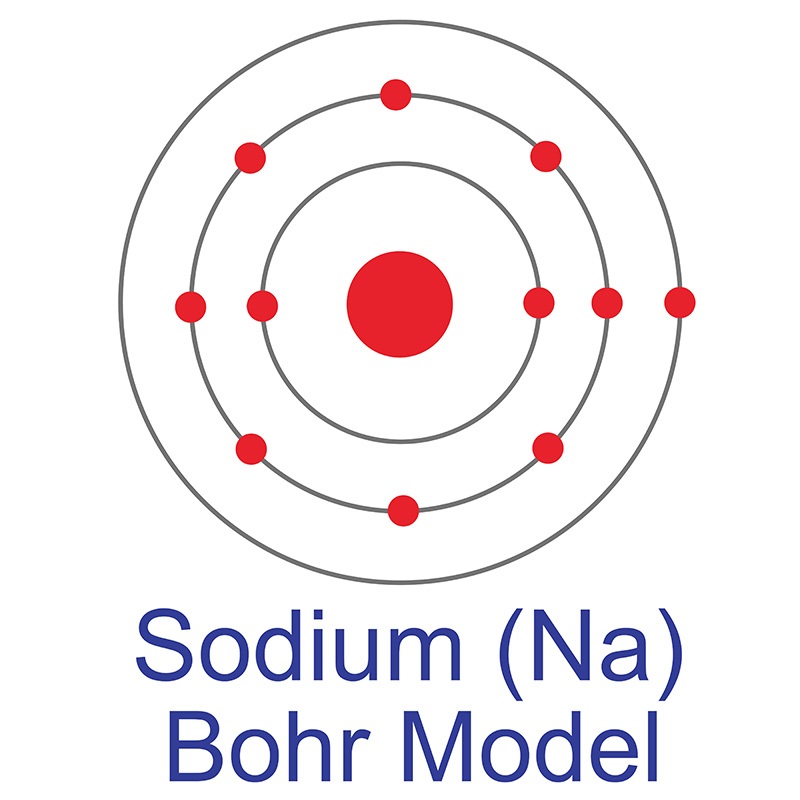 See more Sodium products.
See more Sodium products.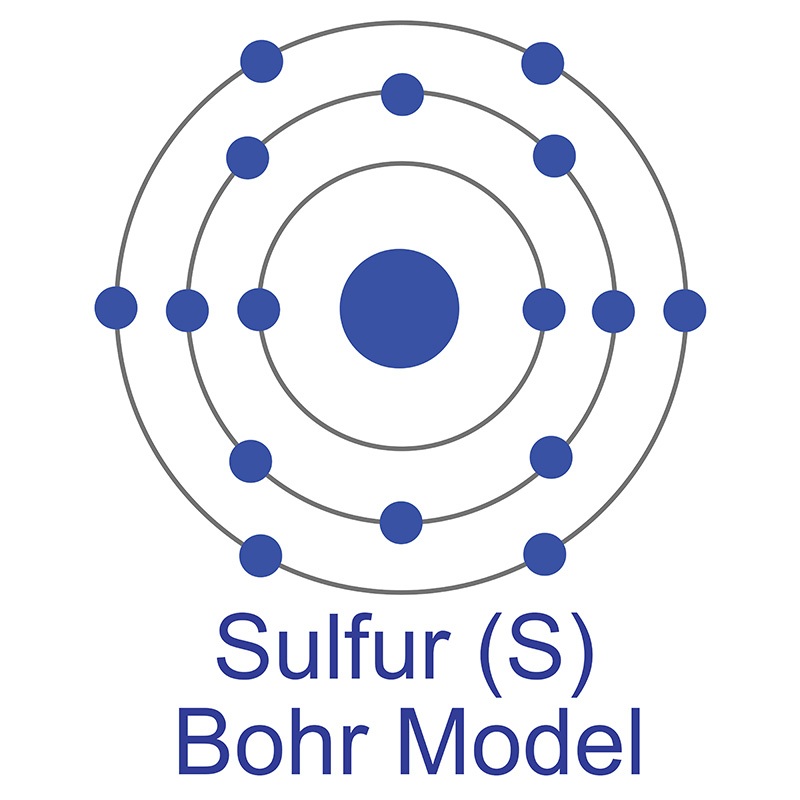 The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.
The number of electrons in each of Sulfur's shells is 2, 8, 6 and its electron configuration is [Ne] 3s2 3p4. In its elemental form, sulfur has a light yellow appearance. The sulfur atom has a covalent radius of 105 pm and a Van der Waals radius of 180 pm. In nature, sulfur can be found in hot springs, meteorites, volcanoes, and as galena, gypsum, and epsom salts. Sulfur has been known since ancient times but was not accepted as an element until 1777, when Antoine Lavoisier helped to convince the scientific community that it was an element and not a compound.
